Collagen Supplementation: The Complete Scientific Guide to What Works, What Doesn’t, and What the Industry Doesn’t Want You to Know
Collagen is the most abundant protein in your body, accounting for roughly 30% of your total protein mass. It provides structural integrity to your skin, bones, tendons, ligaments, cartilage, blood vessels, and the extracellular matrix that holds your tissues together. It is, in the most literal sense, the scaffolding on which you are built. None of that is controversial.
What is controversial, and what has generated a supplement market projected to exceed $9 billion by the end of this decade, is whether swallowing collagen in pill or powder form actually does anything meaningful for that scaffolding. The marketing narrative is simple: your body’s collagen production declines with age (true), this decline contributes to wrinkles, joint pain, and weakening bones (true), and therefore supplementing with collagen can reverse or slow these changes (significantly more complicated than the marketing suggests).
The scientific reality is more interesting than the sales pitch. Collagen supplementation research has produced genuinely promising results in some domains, especially joint health, while revealing serious questions about industry funding bias in others, most notably skin aging. The absorption and metabolism of collagen supplements involves mechanisms that were poorly understood until recently and that challenge basic assumptions about protein digestion. And the collagen supplement market is plagued by a confusion between fundamentally different products, sold under the same name, that work (or don’t work) through entirely different mechanisms.
This analysis examines the full evidence base for collagen supplementation across every major claimed application, from skin and joints to bones and muscle. We address the critical distinction between hydrolyzed collagen peptides and undenatured type II collagen, the absorption question that skeptics and proponents both get partially wrong, and the recent meta-analysis that should change how everyone evaluates collagen’s skin claims. As always, the goal is to give you the evidence, not a sales pitch.
What collagen is and why your body needs it
Collagen biochemistry
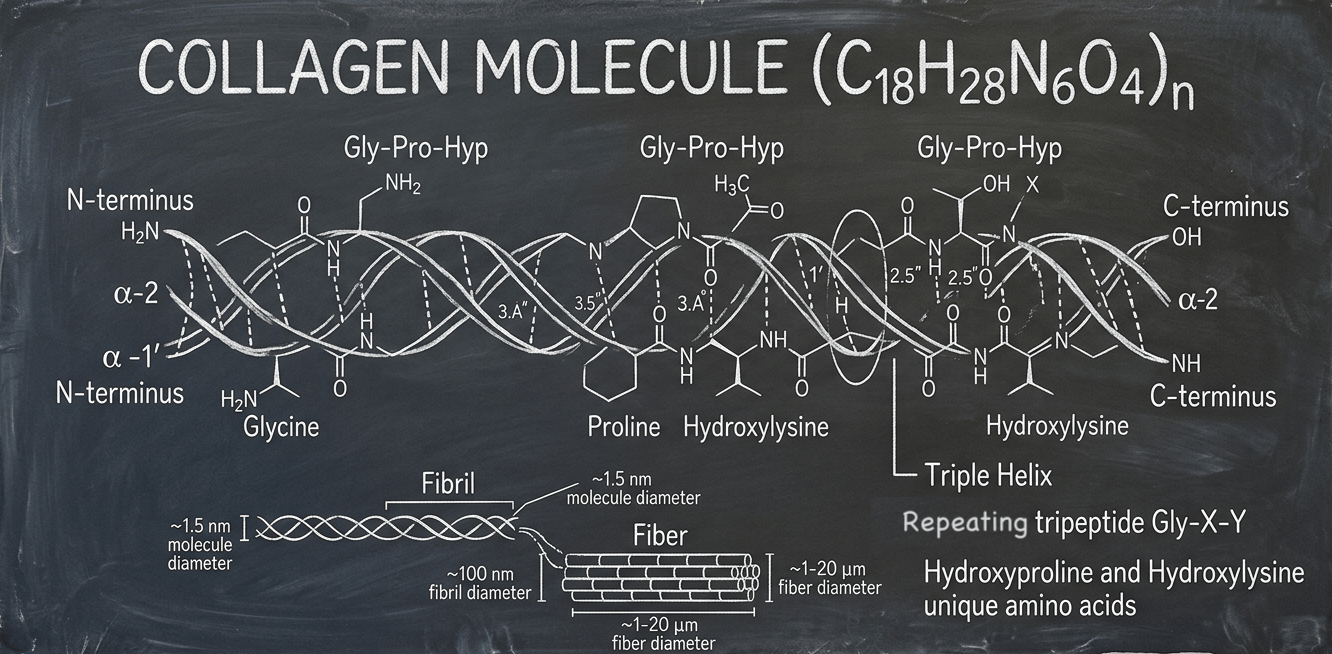
Collagen proteins share a distinctive triple-helix structure: three polypeptide chains wound around each other in a rope-like configuration that gives the protein extraordinary tensile strength. This structure is made possible by collagen’s unusual amino acid composition, which is dominated by glycine (roughly every third residue), proline, and hydroxyproline. Hydroxyproline is particularly notable because it is rare outside of collagen and requires vitamin C for its synthesis, a connection that explains why scurvy (severe vitamin C deficiency) causes collagen to fall apart1Gelse, K., et al. “Collagens: structure, function, and biosynthesis.” Advanced Drug Delivery Reviews, 2003; 55(12): 1531-1546..
Scientists have identified at least 28 distinct collagen types in the human body, though a handful dominate the conversation around supplementation. Type I collagen is by far the most abundant, comprising roughly 90% of the body’s collagen and forming the primary structural component of skin, bones, tendons, and ligaments. Type II collagen is the major collagen of cartilage, where it forms a mesh that traps water-binding molecules called proteoglycans, giving cartilage its cushioning properties. Type III collagen, often found alongside Type I, contributes to the structure of blood vessels, muscles, and organs. Types IV and V play more specialized roles in basement membranes and cell surfaces2Mouw, J.K., et al. “Extracellular matrix assembly: a multiscale deconstruction.” Nature Reviews Molecular Cell Biology, 2014; 15(12): 771-785..
These distinctions matter for supplementation because different collagen types serve different tissues, and, as we will see, the two main categories of collagen supplements (hydrolyzed peptides and undenatured type II) target different collagen types through entirely different biological mechanisms.
The collagen decline problem
Collagen production begins declining around age 25, with the rate of loss accelerating to approximately 1-1.5% per year after age 30. By age 60, most people have lost a substantial fraction of the collagen density in their skin, and the remaining collagen becomes increasingly fragmented and disorganized. This decline is driven by reduced fibroblast activity (the cells that synthesize collagen), increased activity of matrix metalloproteinases (enzymes that break collagen down), cumulative UV damage, oxidative stress, and hormonal changes, particularly the drop in estrogen after menopause3Varani, J., et al. “Decreased collagen production in chronologically aged skin: roles of age-dependent alteration in fibroblast function and defective mechanical stimulation.” American Journal of Pathology, 2006; 168(6): 1861-1868..
In joints, the progressive loss of type II collagen in articular cartilage is a hallmark of osteoarthritis. In bones, collagen degradation contributes to the loss of bone flexibility and resilience that accompanies osteoporosis, because bone is not simply a mineral deposit but a composite material in which hydroxyapatite crystals are deposited within a collagen scaffold. Lose the scaffold, and the mineral alone becomes brittle.
The question that drives the supplement industry is whether exogenous collagen, consumed orally, can meaningfully counteract any of this decline. To answer that, we first need to understand what happens to collagen when you eat it.
The absorption question: what actually happens when you swallow collagen
The skeptic’s objection (and why it’s only half right)
The most common objection to collagen supplements goes something like this: “Collagen is a protein. Your body digests proteins into individual amino acids. Those amino acids don’t know they came from collagen and won’t preferentially rebuild collagen in your skin or joints. You’d get the same result from eating chicken.” This objection reflects legitimate biochemistry from the 20th century, but it is incomplete given what we now know about peptide absorption.
The traditional model of protein digestion holds that dietary proteins are broken down by pepsin in the stomach and pancreatic proteases in the small intestine into individual amino acids, which are then absorbed through amino acid transporters. Under this model, the identity of the original protein is irrelevant once digestion is complete. There is no “collagen” in your bloodstream after eating collagen, just amino acids identical to those from any other protein source.
This model is not wrong. It is, however, incomplete. Research over the past two decades has demonstrated that protein digestion does not always proceed to individual amino acids. Small peptides, primarily di- and tripeptides, are absorbed intact through the peptide transporter PepT1 (SLC15A1) in the small intestine. This is not a minor pathway: peptide absorption is actually faster and more efficient than free amino acid absorption for many peptide sequences4Spanier, B., & Rohm, F. “Proton coupled oligopeptide transporter 1 (PepT1) function, regulation, and influence on the intestinal homeostasis.” Comprehensive Physiology, 2018; 8(2): 843-869..
Collagen-specific peptides in the bloodstream
What makes collagen supplements different from generic protein is the specific peptides that survive digestion. Collagen’s unusual amino acid composition, with its high concentrations of proline and hydroxyproline, produces peptide bonds that are more resistant to standard digestive enzymes than those in most other dietary proteins. The dipeptide prolyl-hydroxyproline (Pro-Hyp) and the tripeptide glycyl-prolyl-hydroxyproline (Gly-Pro-Hyp) are unusually resistant to further breakdown and appear in measurable concentrations in human blood following oral collagen hydrolysate consumption5Iwai, K., et al. “Identification of food-derived collagen peptides in human blood after oral ingestion of gelatin hydrolysates.” Journal of Agricultural and Food Chemistry, 2005; 53(16): 6531-6536..
A 2024 randomized, double-blind crossover study confirmed that collagen hydrolysates from fish, porcine, and bovine sources all produce relevant plasma concentrations of hydroxyproline-containing peptides, regardless of source or molecular weight. Approximately 40% of absorbed hydroxyproline appeared in peptide-bound form rather than as the free amino acid, which means a substantial fraction survives digestion as intact peptides6Virgilio, N., et al. “Absorption of bioactive peptides following collagen hydrolysate intake: a randomized, double-blind crossover study in healthy individuals.” Frontiers in Nutrition, 2024; 11: 1416643..
Even more surprisingly, research using in situ intestinal perfusion has identified collagen-derived peptides larger than tripeptides in portal blood, including peptides up to 15 amino acids long that survive both intestinal and hepatic first-pass metabolism7Osawa, Y., et al. “Absorption and metabolism of orally administered collagen hydrolysates evaluated by the vascularly perfused rat intestine and liver in situ.” Biomedical Research, 2018; 39(1): 1-11..
So the skeptic’s objection needs revision: collagen supplements do more than contribute generic amino acids to the pool. Bioactive peptides do reach the bloodstream intact. The remaining question, and this is where it gets considerably less clear-cut, is whether those circulating peptides actually do anything therapeutically meaningful once they get there. The bioavailability story is more favorable than critics suggest, but bioavailability is not the same as efficacy.
This matters because your body manufactures all of its own collagen. You do not absorb collagen from food and deposit it into your skin or cartilage. Collagen synthesis is an endogenous process that requires glycine, proline, lysine, vitamin C, and a few trace minerals. All of these are readily available from any diet with adequate protein and micronutrient intake. The question is not whether your body can make collagen without a collagen supplement. It can, and it does. The question is whether the specific peptides from hydrolyzed collagen supplements provide a signaling benefit that goes beyond what you would get from eating, say, chicken breast and an orange.
The entire case for hydrolyzed collagen rests on this peptide signaling hypothesis: that Pro-Hyp and related fragments don’t just serve as raw materials but act as molecular signals telling fibroblasts, chondrocytes, and osteoblasts to increase their collagen output. This effect has been demonstrated in cell culture. Whether it occurs at clinically relevant magnitudes in living humans, at the blood concentrations achieved through oral supplementation, is the open question that the clinical trial evidence either confirms or fails to confirm. Keep this framing in mind as we move through the evidence for specific applications, because it explains why some results are positive, some are null, and why funding source matters so much in this research.
Hydrolyzed collagen vs. undenatured collagen: two fundamentally different supplements
Before examining the evidence for specific health claims, you need to understand that “collagen supplements” is an umbrella term covering two products with completely different compositions, doses, and proposed mechanisms of action. Conflating them is like reviewing “vitamin supplements” without distinguishing between vitamin C and vitamin D.
Hydrolyzed collagen (collagen peptides) consists of collagen that has been enzymatically broken down into small peptide fragments, typically 2,000-6,000 daltons. These dissolve easily in liquid and are taken at doses of 2.5-15 grams per day. The proposed mechanism is that absorbed collagen peptides act as signaling molecules, with Pro-Hyp and Gly-Pro-Hyp among the most studied, that stimulate fibroblasts, chondrocytes, and osteoblasts to increase their own collagen production. In other words, the peptides are not building blocks so much as instructions to your cells to build more collagen themselves. Some absorbed amino acids (glycine, proline) may also serve as substrates for new collagen synthesis, though this is likely a secondary mechanism8Pu, S.Y., et al. “Effects of oral collagen for skin anti-aging: a systematic review and meta-analysis.” Nutrients, 2023; 15(9): 2080..
Undenatured type II collagen (UC-II) is a completely different product. It consists of native, intact type II collagen that has not been broken down by heat or enzymes, preserving its three-dimensional structure and antigenic epitopes. It is taken at much lower doses, typically 40 mg per day, and works through a completely different mechanism: oral tolerance. When intact type II collagen reaches the gut-associated lymphoid tissue (Peyer’s patches in the small intestine), it is recognized by the immune system. Through a process called oral tolerization, this exposure trains regulatory T cells to recognize type II collagen as a harmless substance rather than an attack target. Those T cells then circulate to joint tissues, where they suppress the inflammatory immune response that damages cartilage in osteoarthritis9Lugo, J.P., et al. “Undenatured type II collagen (UC-II) for joint support: a randomized, double-blind, placebo-controlled study in healthy volunteers.” Journal of the International Society of Sports Nutrition, 2013; 10(1): 48..
This distinction has practical consequences. Hydrolyzed collagen must be broken down to be effective, so processing (hydrolysis) is a feature, not a bug. Undenatured type II collagen must remain intact to be effective, so any processing that denatures the protein destroys its mechanism of action. Products that claim to contain “Type I, II, III, V, and X collagen” in a single hydrolyzed formula are providing Type II collagen that has lost the structural features required for oral tolerance. It functions only as amino acid substrate at that point, not as an immune modulator10Harris, R.B., et al. “Functional characterization of undenatured type II collagen supplements: are they interchangeable?” Journal of Dietary Supplements, 2022; 19(4): 530-542..
Skin health: the funding problem
What the meta-analyses show (and what they hide)
Skin aging is the primary driver of collagen supplement sales, and until recently, the meta-analytic evidence appeared supportive. A 2023 systematic review of 26 RCTs involving 1,721 participants found that hydrolyzed collagen supplementation significantly improved skin hydration and elasticity compared to placebo11Pu, S.Y., et al. “Effects of oral collagen for skin anti-aging: a systematic review and meta-analysis.” Nutrients, 2023; 15(9): 2080..
Then a 2025 meta-analysis in The American Journal of Medicine did something no previous review had done: it stratified results by funding source and study quality. When all 23 included RCTs were pooled together, collagen supplements significantly improved skin hydration, elasticity, and wrinkles, replicating prior meta-analyses. But when the researchers separated studies by funding source, the picture changed completely. Studies that did not receive pharmaceutical company funding showed no statistically significant effect on skin hydration, elasticity, or wrinkles. Only industry-funded studies showed significant benefits. The same pattern emerged when stratifying by study quality: high-quality studies showed no significant effect, while low-quality studies showed significant improvements in elasticity12Myung, S.K., et al. “Effects of collagen supplements on skin aging: a systematic review and meta-analysis of randomized controlled trials.” American Journal of Medicine, 2025; 138(9): 1264-1277..
This is the first meta-analysis to report no effect of collagen supplements on skin aging, and it did so by asking a question that previous reviews did not: does the answer change depending on who paid for the research?
This does not prove that collagen supplements are useless for skin. It does prove that the evidence base, when subjected to appropriate scrutiny for bias, is far weaker than the marketing would suggest. The positive results that have driven billions in consumer spending come disproportionately from studies funded by the companies selling the products, conducted with methodological shortcomings that inflate effect sizes. Independent, high-quality research has not confirmed those results. If you have read our analysis of evidence-based supplement recommendations, this pattern should be familiar.
What the positive studies actually measured
Even setting aside the funding question, the skin studies raise practical interpretation issues. The outcomes measured, including skin hydration (via corneometer), elasticity (via cutometer), and wrinkle depth (via skin replicas), are objective instrumental measurements, which is methodologically better than subjective self-assessment. However, the clinical significance of the measured improvements is less clear. A statistically significant improvement in cutometer readings does not necessarily translate to a visually noticeable difference in skin appearance.
Most positive studies used doses of 2.5-10 grams of hydrolyzed collagen daily for 8-12 weeks. Common sources included porcine, bovine, and marine (fish) collagen. The subgroup analyses in the 2023 Pu et al. review found no significant differences between collagen sources for skin elasticity outcomes, though marine collagen showed slightly better results for hydration at certain durations. These findings suggest that source, whether bovine, porcine, or marine, is less important than the marketing distinctions between them imply.
Joint health: the strongest evidence domain
Osteoarthritis outcomes
Joint health is where collagen supplementation has produced its most consistent and least conflicted results, with evidence supporting both hydrolyzed collagen peptides and undenatured type II collagen through their different mechanisms.
A 2025 meta-analysis examining collagen supplementation for knee osteoarthritis pooled data from 11 RCTs involving 870 participants. The analysis found significant improvements in both function (WOMAC total score) and pain scores favoring collagen supplementation over placebo. The improvements were statistically and clinically meaningful, but heterogeneity between studies was high13Simental-Mendía, M., et al. “Effect of collagen supplementation on knee osteoarthritis: an updated systematic review and meta-analysis of randomised controlled trials.” Clinical and Experimental Rheumatology, 2025; 43(1): 126-134..
The UC-II evidence stands out. A landmark 2016 randomized, double-blind, placebo-controlled trial compared 40 mg of UC-II daily to both placebo and a glucosamine-chondroitin combination (1,500 mg/1,200 mg daily) in 191 patients with knee osteoarthritis over 180 days. UC-II produced significantly greater reductions in WOMAC pain and function scores than both placebo and glucosamine-chondroitin. This finding was clinically significant because glucosamine-chondroitin is one of the most widely used joint health supplements, and UC-II outperformed it at a fraction of the dose14Lugo, J.P., et al. “Efficacy and tolerability of an undenatured type II collagen supplement in modulating knee osteoarthritis symptoms: a multicenter randomized, double-blind, placebo-controlled study.” Nutrition Journal, 2016; 15: 14..
However, a 2025 RCT published in Scientific Reports tested a combined UC-II plus hydrolyzed collagen supplement against placebo in 68 knee osteoarthritis patients over 12 weeks and found no significant differences between groups in pain, function, rescue medication use, or patient satisfaction. Both groups improved, but the collagen group did not improve more than placebo15Yuenyongviwat, V., et al. “Efficacy of combined undenatured type II collagen and hydrolysed collagen supplementation in knee osteoarthritis: a randomised controlled trial.” Scientific Reports, 2025; 15: 32313..
This mixed picture, with some positive and some null results, is common in supplement research and suggests that benefits may depend on factors like disease severity, collagen type and dose, study duration, and patient characteristics. The overall weight of evidence leans positive for joint health, but it is not unanimous.
Exercise-related joint discomfort
For healthy individuals without diagnosed osteoarthritis who experience joint discomfort during exercise, the evidence is more limited. The 2013 Lugo et al. study in healthy volunteers found that 40 mg of UC-II daily for 120 days improved knee extension and extended the time subjects could exercise before experiencing joint pain16Lugo, J.P., et al. “Undenatured type II collagen (UC-II) for joint support: a randomized, double-blind, placebo-controlled study in healthy volunteers.” Journal of the International Society of Sports Nutrition, 2013; 10(1): 48..
Hydrolyzed collagen peptides at doses of 5-10 grams daily have shown some benefit for exercise-related joint pain in athletes, with the clearest benefits appearing in studies lasting 12 weeks or longer. The proposed mechanism involves stimulation of chondrocyte activity and enhanced collagen synthesis in cartilage and connective tissue. A 2024 systematic review of collagen peptide supplementation and musculoskeletal performance found modest positive effects on muscle function and some improvements in joint pain scores. The authors noted that most studies were small and many had methodological limitations17Kirmse, M., et al. “Collagen peptide supplementation and musculoskeletal performance: a systematic review and meta-analysis.” Deutsche Zeitschrift für Sportmedizin, 2024; 75: 179-188..
Bone health
Bone is approximately 90% type I collagen by organic mass, and the collagen matrix provides the framework for mineral deposition. The rationale for collagen supplementation to support bone health is mechanistically sound, and the early evidence is promising if still limited.
A 2025 meta-analysis of RCTs examining collagen peptide supplementation found significant increases in bone mineral density at both the femoral neck and spine, along with improvements in bone turnover markers and muscle function. The improvements were more pronounced when collagen was combined with calcium and vitamin D, which the authors interpreted as evidence of synergistic effects18Sun, C., et al. “Efficacy of collagen peptide supplementation on bone and muscle health: a meta-analysis.” Frontiers in Nutrition, 2025; 12: 1646090..
The most cited individual study is a 2018 RCT by König et al. in postmenopausal women with osteopenia, which found that 5 grams of specific collagen peptides daily for 12 months significantly increased bone mineral density at the femoral neck and lumbar spine, with concurrent improvements in bone formation markers and reduction of a bone degradation marker. These results are encouraging, but the study was relatively small (131 participants) and was funded by the collagen peptide manufacturer19König, D., et al. “Specific collagen peptides improve bone mineral density and bone markers in postmenopausal women: a randomized controlled study.” Nutrients, 2018; 10(1): 97..
Given the skin research findings about funding bias, the bone health results, which come largely from industry-sponsored studies, warrant cautious optimism rather than firm conclusions. Independent replication in larger trials is needed.
Muscle mass and athletic performance
Collagen is not an ideal protein source for muscle building. Its amino acid profile is poor compared to whey, casein, or egg protein, with minimal leucine content (the primary trigger for muscle protein synthesis) and a complete absence of tryptophan, making it an incomplete protein. If your primary goal is muscle hypertrophy, creatine and a complete protein source will serve you far better than collagen.
Collagen peptides may, however, play a role in connective tissue adaptation to exercise. Tendons, ligaments, and the extracellular matrix of muscle are collagen-rich structures that adapt to training loads. Small studies have found that collagen peptide supplementation (15 grams) combined with resistance training can increase collagen synthesis in tendons and improve tendon mechanical properties. Whether these changes translate to meaningful injury prevention remains unproven20Nulty, C.D., et al. “Hydrolyzed collagen supplementation prior to resistance exercise augments collagen synthesis in a dose-response manner in resistance-trained, middle-aged men.” American Journal of Physiology: Endocrinology and Metabolism, 2024; 327: E668-E677..
For older adults concerned about sarcopenia (age-related muscle loss), a few studies have found that collagen peptide supplementation combined with resistance training produced greater gains in fat-free mass and muscle strength compared to resistance training with placebo. These results may reflect collagen’s effects on the connective tissue components of muscle rather than on myofibrillar protein itself.
Sources, types, and what the labels mean
Marine collagen
Marine collagen, derived primarily from fish skin and scales, is marketed as superior due to claims of higher bioavailability. The bioavailability claim rests on the fact that marine collagen is typically hydrolyzed to a lower molecular weight (2,000-3,000 daltons) than many bovine products (3,000-8,000 daltons). This is a processing difference, not an inherent property of fish collagen itself. When bovine and marine collagen are hydrolyzed to the same molecular weight, the 2024 crossover bioavailability study found comparable absorption profiles21Virgilio, N., et al. “Absorption of bioactive peptides following collagen hydrolysate intake: a randomized, double-blind crossover study in healthy individuals.” Frontiers in Nutrition, 2024; 11: 1416643..
Marine collagen provides predominantly type I collagen, making it relevant for skin and bone applications but not for the UC-II joint health mechanism. It is also more expensive per gram than bovine collagen and may have a fishy taste or odor that some users find objectionable. For consumers choosing between marine and bovine hydrolyzed collagen, the difference is primarily one of personal preference, dietary restrictions (marine is suitable for pescatarians), and cost, not efficacy.
Bovine collagen
Bovine collagen, sourced from cow hides and bones, is the most common and least expensive collagen supplement. It provides primarily types I and III collagen. Most of the clinical research on hydrolyzed collagen for skin, bone, and connective tissue health has used bovine-derived products. Bovine collagen from bone broth also contains small amounts of type II collagen, though not in the undenatured form required for the oral tolerance mechanism.
Chicken collagen
Chicken sternum cartilage is the primary source for UC-II (undenatured type II collagen) products. The patented low-temperature extraction process preserves the native triple-helix structure and antigenic epitopes needed for the oral tolerance mechanism. Chicken collagen is also used in some hydrolyzed products, but its primary relevance is for UC-II joint health applications.
The “multi-collagen” marketing problem
Products marketed as containing “Types I, II, III, V, and X collagen” in a single formula deserve particular skepticism. No clinical trial has ever demonstrated that combining multiple collagen types in one product produces better outcomes than a single-type supplement at a studied dose. The marketing claim “5 types in one scoop” is exactly that: a marketing claim, not a research-backed advantage.
Worse, the multi-type approach creates a mechanical contradiction. UC-II requires intact, undenatured protein to work through oral tolerance. The hydrolysis process that makes types I and III absorbable as peptides destroys the type II structure that enables immune modulation. A product that hydrolyzed all five types together delivers type II as nothing more than amino acids, eliminating the very mechanism that makes it interesting for joint health. If you want the benefits of both hydrolyzed collagen peptides and UC-II, you need two separate products, not one blended powder.
Dosing, timing, and practical considerations
For hydrolyzed collagen peptides: Clinical studies have used doses ranging from 2.5 to 15 grams per day, with 5-10 grams being the most common effective dose range. The research does not strongly support one timing over another, though some evidence suggests that consuming collagen peptides 30-60 minutes before exercise (with vitamin C to support collagen synthesis) may enhance connective tissue adaptation. Collagen peptides dissolve easily in hot or cold liquids and are generally well-tolerated, with minimal reported side effects beyond occasional mild GI discomfort.
For UC-II (undenatured type II collagen): The studied dose is 40 mg per day, taken once daily. Some practitioners recommend taking it on an empty stomach to optimize interaction with gut-associated lymphoid tissue, though this has not been rigorously tested. The dose is low enough that it adds negligible calories or protein to the diet.
Duration expectations: Collagen is not a rapid-onset supplement. Most positive studies required 8-12 weeks of daily supplementation before measuring significant outcomes, with some joint studies running 24 weeks. Expecting noticeable results in days or weeks is unrealistic and inconsistent with the biology of collagen turnover in tissues.
Co-factors: Collagen synthesis requires vitamin C as a cofactor for prolyl and lysyl hydroxylase, the enzymes that modify proline and lysine residues in newly synthesized collagen. Supplementing collagen without adequate vitamin C status is like providing building materials without mortar. Zinc and copper also play roles in collagen cross-linking and maturation.
Safety and quality considerations
Collagen supplements have an excellent overall safety profile, with no serious adverse events reported in clinical trials. The most common side effects are mild GI symptoms (bloating, fullness, unpleasant taste) that typically resolve with continued use. Unlike biotin, which causes potentially dangerous laboratory test interference at high doses, collagen supplements have no known interactions with clinical assays.
Quality concerns center on sourcing and contamination. Collagen derived from animal connective tissues can potentially contain heavy metals, with marine-sourced products carrying higher risk. Third-party testing through organizations like NSF, USP, or independent laboratories provides reasonable assurance of purity and label accuracy. Consumers should verify that products state the collagen source (bovine, marine, chicken), the collagen type(s), the degree of hydrolysis (for peptide products), and ideally third-party testing certification.
Individuals with allergies to fish, shellfish, eggs, or chicken should check collagen supplement sourcing carefully. Bovine collagen is generally the safest choice for individuals with seafood or poultry allergies. Collagen supplements are not suitable for vegans or strict vegetarians, as all currently available collagen is animal-derived. (Products marketed as “vegan collagen” typically contain amino acids and cofactors intended to support the body’s own collagen production, not actual collagen protein.)
Conclusion
Collagen supplementation is more scientifically interesting than its marketing suggests and less proven than its proponents claim. The absorption story, once considered the supplement’s fatal flaw, turns out to be more favorable than classical protein digestion models predicted, with bioactive peptides genuinely reaching the bloodstream in intact form. The distinction between hydrolyzed collagen peptides and undenatured type II collagen represents a real biological difference with meaningful practical implications that most consumers and many retailers do not understand.
The evidence is strongest for joint health, where both hydrolyzed collagen and UC-II have produced positive results in multiple RCTs, with UC-II outperforming glucosamine-chondroitin in head-to-head comparison. The evidence is weakest, and most compromised by funding bias, for skin anti-aging, where the 2025 American Journal of Medicine meta-analysis found that independent, high-quality studies showed no significant benefit. Bone health and connective tissue applications show promise but lack the independent replication needed for confident recommendations.
For consumers weighing whether to invest in collagen supplementation: if joint health is your primary concern, UC-II at 40 mg daily has the strongest independent evidence base and is relatively inexpensive. If you are interested in skin or bone applications, hydrolyzed collagen peptides at 5-10 grams daily are reasonable to try, but expectations should be calibrated to the reality that the strongest positive evidence comes from industry-funded research. Avoid “multi-collagen” products that claim to combine every type in one formula, as this approach has no clinical evidence and creates a mechanical contradiction for the UC-II mechanism. Ensure adequate vitamin C intake regardless of which form you choose.
The collagen supplement market thrives on the gap between what consumers hope these products will do and what the research has confirmed they do. Closing that gap, one evidence review at a time, is what this site exists for.
Generate Your Stack. Avoid Conflicts. Optimize Absorption.
Most supplement advice ignores timing, interactions, and dose-response curves. Smart Stack Builder doesn't. Get a personalized daily schedule with interaction flags and dosage guidance, built on the same evidence base as this article.
References
- 1
- 2
- 3
- 4
- 5
- 6Virgilio, N., et al. “Absorption of bioactive peptides following collagen hydrolysate intake: a randomized, double-blind crossover study in healthy individuals.” Frontiers in Nutrition, 2024; 11: 1416643.
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
- 15
- 16
- 17
- 18
- 19
- 20
- 21
This article is part of our Supplements hub: A science-backed collection of ingredient research, clinical dosage reviews, and optimized stack insights.