The NSF certification mark shows up on supplement labels without much fanfare. It’s small, it’s not flashy, and most consumers walk right past it. Meanwhile, the front of the bottle is doing the real selling with bold claims about potency and purity.
But in an industry where companies can put almost anything on a label without proving it’s true before the product hits shelves, that quiet little NSF mark is one of the few indicators that someone independent actually checked 1U.S. Food and Drug Administration. “Dietary Supplements: What You Need to Know.” FDA Consumer Health Information, 2022..
Dietary supplements in the United States don’t require pre-market approval for safety or efficacy. The Dietary Supplement Health and Education Act of 1994 (DSHEA) put the regulatory burden largely on the FDA to prove a product is unsafe after it’s already being sold, rather than requiring manufacturers to prove safety before going to market 2Dietary Supplement Health and Education Act of 1994, Public Law 103-417, 103rd Congress.. That framework means contaminated and mislabeled products reach consumers regularly. Third-party certification programs like NSF exist because the default regulatory environment doesn’t catch most of these problems before they become someone else’s health issue.
This article covers what NSF certification actually involves at a technical level, what the different certification tiers mean, how NSF compares to other certification programs, and who benefits most from paying attention to it.
What NSF International actually is
NSF International started as the National Sanitation Foundation, established in 1944 to address food safety and sanitation problems during post-war industrial expansion 3NSF International. “History and Mission: 75 Years of Public Health Protection.” Organizational Documentation, 2019.. The original focus was developing standardized public health codes for foodservice environments, back when contamination risks were poorly understood and inconsistently regulated across states.
Over the decades that followed, NSF expanded into water treatment, food equipment, plumbing, building materials. The through-line was always the same kind of problem: situations where quality failures are invisible to the end user but the consequences aren’t. The organization operates as an independent nonprofit, not a government agency or a commercial testing company, which matters when you’re evaluating how much weight to give their stamp.
The move into dietary supplement certification came during the supplement boom that followed DSHEA’s passage. Supplement sales were growing fast, regulatory oversight wasn’t keeping pace, and there was an obvious gap between what consumers assumed about product quality and what was actually being verified 4Cohen PA. “American roulette—contaminated dietary supplements.” New England Journal of Medicine. 2009;361(16):1523-1525.. NSF already had the analytical chemistry infrastructure and the institutional independence to fill that gap. Their laboratories were equipped for high-precision contaminant detection and ingredient verification from decades of work in other industries.
Today, NSF operates in over 180 countries, maintains ISO-accredited laboratories worldwide, and participates in developing more than 90 public health standards across multiple industries 5NSF International. “Global Operations and Accreditation Status.” Annual Report, 2023..
How NSF certification works
NSF doesn’t just test a bottle of supplements and call it a day. The certification process involves multiple layers of verification, each targeting a different way products can go wrong.

NSF Contents Certified (standard certification)
This is the baseline tier, designed for general supplement consumers who want some assurance that what’s on the label is what’s in the bottle.
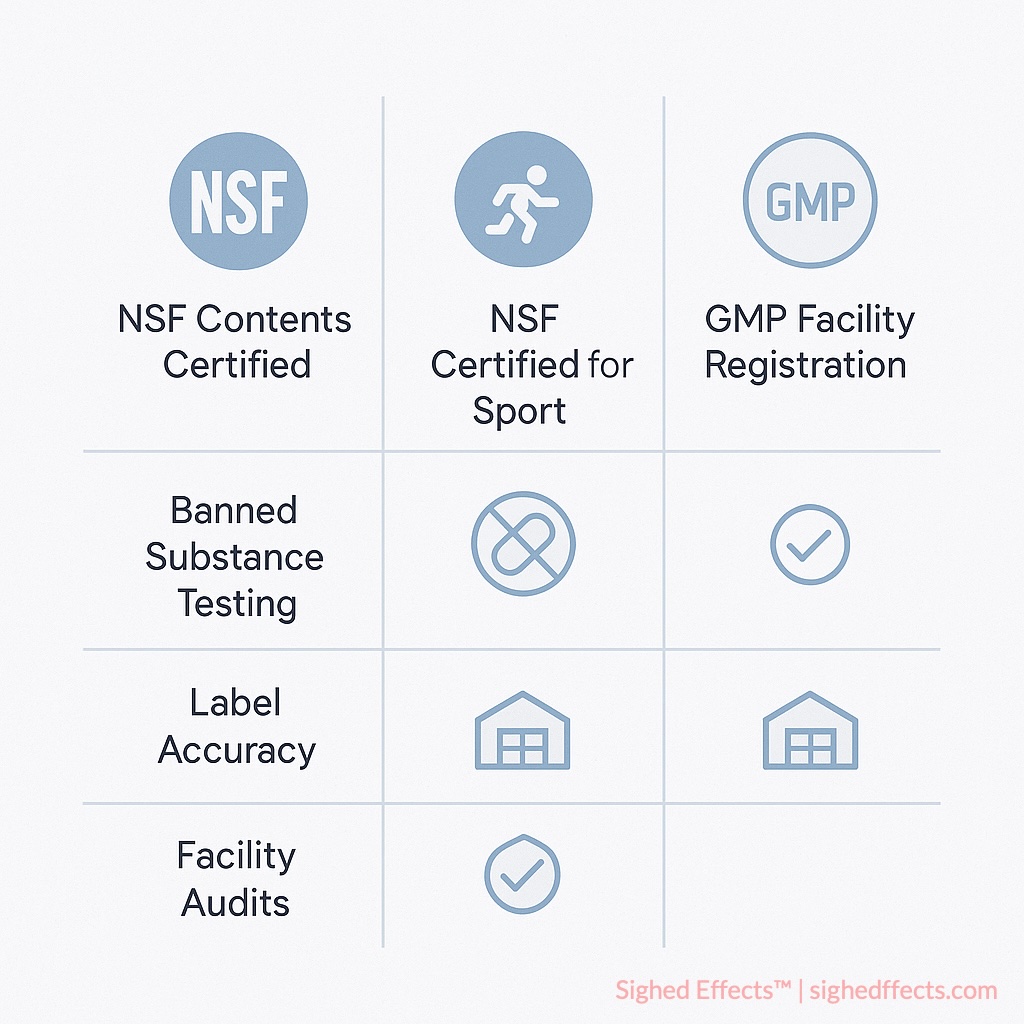
Ingredient identity and potency verification is where it starts. Chemical analysis confirms that the labeled compounds match actual contents. Quantitative testing checks whether ingredient concentrations fall within specified tolerances. For botanical supplements, species authentication uses DNA analysis to confirm you’re actually getting the plant the label claims. Stability testing verifies that potency holds up through the product’s shelf life, not just at the time of manufacture.
Contaminant screening covers heavy metals (lead, mercury, cadmium, arsenic) using inductively coupled plasma mass spectrometry (ICP-MS), microbiological testing for pathogenic bacteria, yeast, and mold, pesticide residue screening for botanical ingredients, and residual solvent testing to catch manufacturing process contaminants.
Manufacturing quality assurance rounds it out. NSF audits facilities against Good Manufacturing Practice (GMP) standards, reviews supply chain documentation for ingredient traceability, evaluates quality control systems, and validates batch records and testing procedures.
NSF Certified for Sport
This is the tier that gets the most attention, and for good reason. It builds on everything in the standard certification and adds screening for substances banned in competitive athletics.

NSF Certified for Sport tests for over 290 substances on the World Anti-Doping Agency (WADA) Prohibited List and those banned by major professional sports leagues 6NSF International. “Certified for Sport Program.” nsfsport.com. Accessed 2025.. That includes anabolic agents, stimulants, hormone modulators, diuretics, masking agents, and emerging designer compounds. The screening isn’t just a checkbox exercise; it’s calibrated to detect trace contamination at levels that could trigger a positive result on athletic drug testing.
The testing requirements are also more rigorous at the batch level. Individual lots get tested before they’re released to market. NSF conducts ongoing shelf audits by pulling products from retail locations. Unannounced facility inspections happen after certification is granted. And there’s adverse event tracking built into the program so that problems that emerge post-certification get flagged and investigated.
Marketing materials also get reviewed under this program. NSF checks that claims on labels and promotional content comply with anti-doping standards, that ingredient disclosure meets or exceeds standard labeling requirements, and that batch number traceability systems are in place for contamination investigation if something goes wrong.
Facility auditing

The facility audit process goes deeper than standard GMP inspections. NSF evaluates cross-contamination prevention systems, raw material storage conditions, environmental monitoring for temperature, humidity, and air quality, and waste management and cleaning validation. They also look at personnel training documentation, standard operating procedure implementation, quality assurance team qualifications, and corrective action protocols.
On the documentation side, NSF verifies batch record accuracy and retention, supplier qualification and monitoring programs, certificates of analysis, and product recall procedures. The point is to evaluate the entire system that produces the supplement, not just the finished product sitting in a bottle.
The analytical science behind it
The testing methods NSF uses are adapted from pharmaceutical and food safety standards, applied specifically to dietary supplement challenges. These methods get updated regularly as new contaminants emerge and analytical technology improves.
NSF laboratories use liquid chromatography-mass spectrometry (LC-MS/MS) and ICP-MS for compound identification and quantification 7Association of Official Analytical Chemists. “Guidelines for Single Laboratory Validation of Chemical Methods.” AOAC Guidelines, 2022.. LC-MS/MS is particularly useful for banned substance detection because it can identify compounds at parts-per-billion concentrations, which matters when even trace contamination can end an athlete’s career.
Method validation involves reference standard materials for accurate quantification, blank sample analysis to confirm the lab itself isn’t introducing contamination, duplicate sample testing for measurement precision, and proficiency testing through inter-laboratory comparison programs. Banned substance detection thresholds are aligned with WADA technical requirements. Heavy metal limits reflect current toxicological safety data. Microbial testing uses both culture-based and molecular detection methods.
Where contamination comes from
NSF’s testing protocols are designed around the specific pathways that contamination actually enters supplement products:
Raw materials are one of the most common sources. Suppliers may provide ingredients that are cross-contaminated during processing or storage. Agricultural inputs can carry pesticide and herbicide residues. Certificates of analysis from suppliers aren’t always reliable, which is why NSF does independent confirmation testing rather than taking documentation at face value.
Manufacturing processes introduce contamination when equipment is shared between different products without adequate cleaning validation. This is how banned substances end up in supplements that don’t intentionally contain them. A facility that produces a testosterone booster on Monday and a protein powder on Tuesday can cross-contaminate the protein powder if cleaning protocols aren’t rigorous enough.
Post-manufacturing contamination is less discussed but still real. Packaging materials can leach compounds. Storage and distribution conditions affect product stability. This is why NSF’s monitoring includes shelf-life stability studies and retail sampling programs, not just testing at the factory door.

How NSF compares to other certification programs
NSF isn’t the only option. Understanding what each program actually tests for (and what it doesn’t) matters for making a useful choice.
NSF vs. USP
USP (United States Pharmacopeia) Verified certification comes from a pharmaceutical standards tradition. It emphasizes ingredient identity, potency accuracy, and dissolution testing for tablets and capsules 8United States Pharmacopeia. “USP Verified Dietary Supplements Program.” Quality Standards Documentation, 2023.. USP is strong on fundamental quality parameters. Where it differs from NSF is scope: USP doesn’t include banned substance screening or athlete-specific compliance testing.
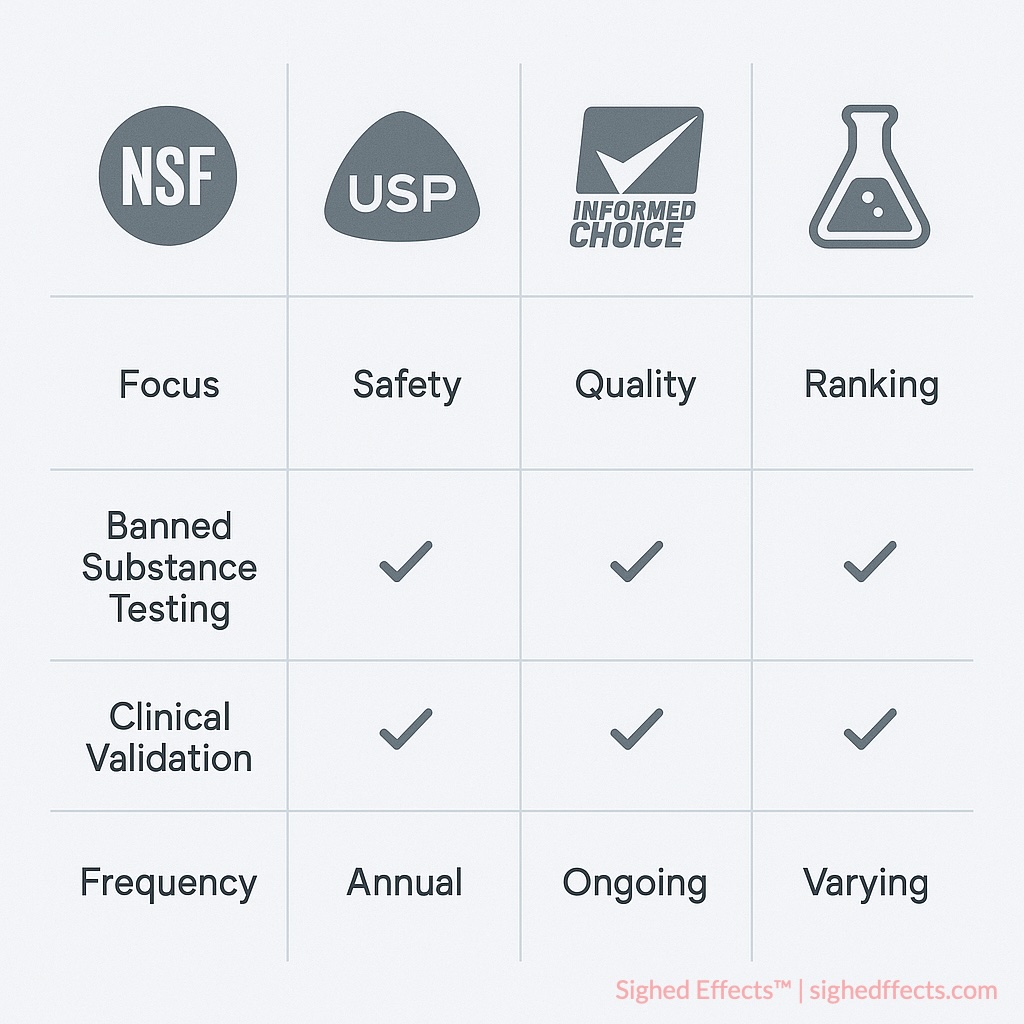
Both organizations are ISO-accredited and participate in international standards development. USP standards are referenced in FDA regulations and pharmacopoeial monographs. NSF Certified for Sport is recognized by USADA, the NCAA, and professional sports leagues. Neither certification evaluates clinical efficacy or therapeutic outcomes.
If you’re a competitive athlete subject to drug testing, NSF Certified for Sport is the more relevant program because it specifically addresses banned substance exposure. If you’re a general consumer primarily concerned with whether your multivitamin contains what the label says it does, both NSF and USP provide solid quality verification.
NSF vs. Informed Choice
Informed Choice is a global banned substance testing program run by LGC Group, with particular strength in European and international markets Informed Choice is a global banned substance testing program run by LGC Group, with particular strength in European and international markets 9LGC Group. “Informed Choice Certification Program.” choice.wetestyoutrust.com. Accessed 2025..
The programs differ in testing frequency and scope. NSF requires lot-by-lot testing before product release; Informed Choice emphasizes periodic monitoring through monthly batch testing. NSF includes full label accuracy verification alongside banned substance screening; Informed Choice focuses primarily on the banned substance side. NSF maintains publicly searchable databases with batch-specific information, which gives consumers a way to verify certification claims independently. Informed Choice has broader international recognition, particularly in markets outside North America.
For competitive athletes, both programs provide adequate anti-doping protection. The choice often comes down to what’s available for your specific supplement and whether international distribution matters.
Independent lab testing without formal certification
Some supplement companies hire independent laboratories for product testing without going through a formal certification program. This can provide useful quality data, but it has structural limitations. There are no standardized testing protocols or detection limit requirements. There’s no facility auditing or manufacturing process evaluation. The manufacturer chooses which lab to use, which introduces potential conflicts of interest. And there’s no ongoing monitoring or post-market surveillance.
Formal certification programs address these problems because standardized methodologies and independent oversight are baked in, and the monitoring doesn’t stop after the initial test. Ad hoc testing can supplement formal certification, but it’s not a substitute for it.
Who benefits from NSF certification
Athletes in tested sports

Professional sports leagues including the NFL, MLB, NHL, and NBA recognize NSF Certified for Sport as an acceptable risk mitigation strategy 10International Testing Agency. “Supplements.” ITA Athlete Hub. ita.sport/athlete-hub/supplements/. Accessed 2025.. Team nutritionists and medical staff frequently limit approved supplements to certified products. The stakes here are not ambiguous: a positive drug test from contaminated supplements can end a career, and “I didn’t know it was in there” is not a defense that anti-doping agencies accept.
NCAA compliance officers recommend certified products for similar reasons. Scholarship eligibility is on the line, and institutional liability concerns push universities toward policies that require verified products. USADA explicitly references NSF Certified for Sport in its athlete education materials, and elite training centers typically stock only certified products.
If you’re subject to any form of drug testing, using non-certified supplements is an unforced risk.
Healthcare providers
Physicians and other practitioners face a difficult position with supplements. Patients ask for recommendations, but the quality variability across the supplement market makes blanket endorsements risky 11Kadro ZO, Chilcoat A, Hill J, Kenney S, Nguyen C, Post E, Corbett AH, Asher GN, Faurot K. Healthcare Professionals’ Perspectives on Improving Dietary Supplement Documentation in the Electronic Medical Record: Current Challenges and Opportunities to Enhance Quality of Care and Patient Safety. Glob Adv Integr Med Health. 2023 Dec 19;12:27536130231215029. doi: 10.1177/27536130231215029. PMID: 38130392; PMCID: PMC10734342.. NSF certification gives clinicians a systematic way to filter for quality without having to personally evaluate manufacturing processes.
Hospital systems are starting to develop formularies that prioritize certified products, and outpatient clinics are increasingly recommending certified brands. Clinical research is moving in this direction too: supplement intervention trials are beginning to specify certified products to ensure consistency across study sites, which matters for reproducibility.
General consumers
For someone not subject to drug testing or clinical oversight, the situation is different. Certification does provide real quality assurance, but the premium pricing (certified products typically cost 15-30% more than non-certified alternatives) may or may not be justified depending on the supplement, how often you take it, and your individual risk tolerance.
Where certification matters most for general consumers is with daily-use foundational supplements: multivitamins, protein powders, creatine, omega-3s. These are products you take repeatedly over long periods, and any contamination or potency problem compounds with chronic exposure. For something you take occasionally or at low doses, the risk profile changes.
The business side
What certification costs manufacturers
Pursuing NSF certification isn’t cheap, and the costs go beyond lab fees. There are direct costs: application and documentation review fees, laboratory testing across full analytical panels, facility audit expenses, and ongoing monitoring fees. But the indirect costs are often larger. Manufacturers may need to modify production processes, implement enhanced documentation and quality control systems, upgrade supply chain transparency, and invest in marketing that communicates why certification matters.
The process can take anywhere from several months to over a year to implement fully, depending on how much a manufacturer’s existing systems need to change.
What it means for market dynamics
Certified products tend to command price premiums of 20-35% over non-certified alternatives. Professional and institutional sales channels favor certified products. Military and government contracts increasingly specify certification requirements.
The flip side is that certification costs create barriers to entry. Smaller manufacturers with limited capital may produce quality products but lack the resources to pursue formal certification. This can concentrate the market among larger brands, which isn’t necessarily a quality signal on its own. A small company that can’t afford NSF certification isn’t automatically making an inferior product. They just haven’t undergone the verification process.
Regulatory context
The FDA references NSF certification in enforcement actions, which tells you something about how regulators view these programs even if they’re technically voluntary 12U.S. Food and Drug Administration. “FDA Issues Warning About Certain Supplements Substituted with Toxic Yellow Oleander.” FDA Alerts, Advisories & Safety Information, 2023.. FDA investigators have cited the absence of third-party testing in warning letters to manufacturers. Import inspections may factor certification status into risk assessments.
NSF certification works alongside FDA oversight rather than replacing it. The FDA sets the regulatory floor; NSF certification exceeds it. This matters because the regulatory floor for supplements is low. Until Congress raises it (and there have been ongoing discussions about mandatory third-party testing for certain product categories), voluntary certification programs remain the primary mechanism for quality assurance above the legal minimum.
What certification doesn’t tell you
NSF certification’s limitations matter as much as its strengths, and the most important one is this: NSF verifies that a product is safe, contains what the label says, and is free from contaminants. It says nothing about whether the ingredients actually work. Efficacy evaluation is completely outside NSF’s scope. A certified product can be pure, accurately labeled, and still useless for its intended purpose if the ingredient lacks clinical evidence at the dose provided.
Coverage is another constraint. NSF certification covers a small fraction of available supplement products, and many product categories have few or no certified options. If your preferred supplement doesn’t have a certified alternative, you’re back to evaluating brands through other quality indicators.
On the analytical side, detection limits are real. NSF’s methods achieve high sensitivity, but emerging contaminants and novel synthetic compounds may escape detection until testing protocols get updated. And lot-by-lot testing, while better than batch sampling, still can’t catch every intermittent contamination event. Manufacturing changes between certification periods, supply chain disruptions, and seasonal variation in raw materials can all affect product quality in ways that periodic testing won’t fully capture. NSF’s ongoing monitoring helps, but it doesn’t make the process airtight.
Verifying certification claims

NSF maintains public databases where you can look up individual products and confirm their certification status. When batch-specific information is available, you can verify that the particular lot you purchased was tested. Certification has expiration dates and renewal requirements, so checking periodically for products you use regularly is worth the minor effort. If you encounter a product displaying the NSF mark that doesn’t appear in their database, you can report it to NSF and to the FDA.
Where this is heading
Analytical technology keeps advancing. There’s been interest in blockchain-based traceability and predictive quality modeling, and QR codes on packaging that link directly to batch-specific test results are already in use by some certified brands. Whether any of these technologies improve on existing processes in practice, or just add marketing polish, remains to be seen.
On the regulatory side, the conversation about mandatory third-party testing for supplements keeps resurfacing in Congress and at the FDA. If mandatory requirements do come, organizations like NSF that already have the infrastructure and methodology in place will be positioned to support expanded oversight. Whether that actually happens is a political question as much as a scientific one.
The supplement market keeps growing. The gap between what consumers assume about product quality and what anyone has actually verified remains wide. NSF certification doesn’t close that gap entirely, but for consumers willing to look past the marketing on the front of the bottle, it’s one of the few tools that holds up to scrutiny.
