Should You Trust In-House Supplement Testing? A Deep Look at Internal Quality Control

In-house lab testing: what it does, what it misses, and how to evaluate it
Most supplement companies do some form of internal testing. Under FDA regulations (21 CFR Part 111), dietary supplement manufacturers are required to verify the identity of incoming ingredients, confirm that finished products meet their specifications, and maintain documentation of these checks as part of current Good Manufacturing Practice (cGMP) compliance.1U.S. Food and Drug Administration. 21 CFR Part 111 — Current Good Manufacturing Practice in Manufacturing, Packaging, Labeling, or Holding Operations for Dietary Supplements. https://www.ecfr.gov/current/title-21/chapter-I/subchapter-B/part-111 This internal quality control is a normal and necessary part of supplement manufacturing.
The question isn’t whether in-house testing happens. It’s whether in-house testing alone tells you enough about what’s in the product you’re buying. The answer, in most cases, is that it doesn’t, not because the testing is fraudulent, but because it’s structurally limited in ways that matter.
This article covers what in-house labs actually do during production, where the capability gaps are, how to evaluate brands that reference their internal testing, and how in-house QC relates to the independent testing and certification programs covered elsewhere on this site.

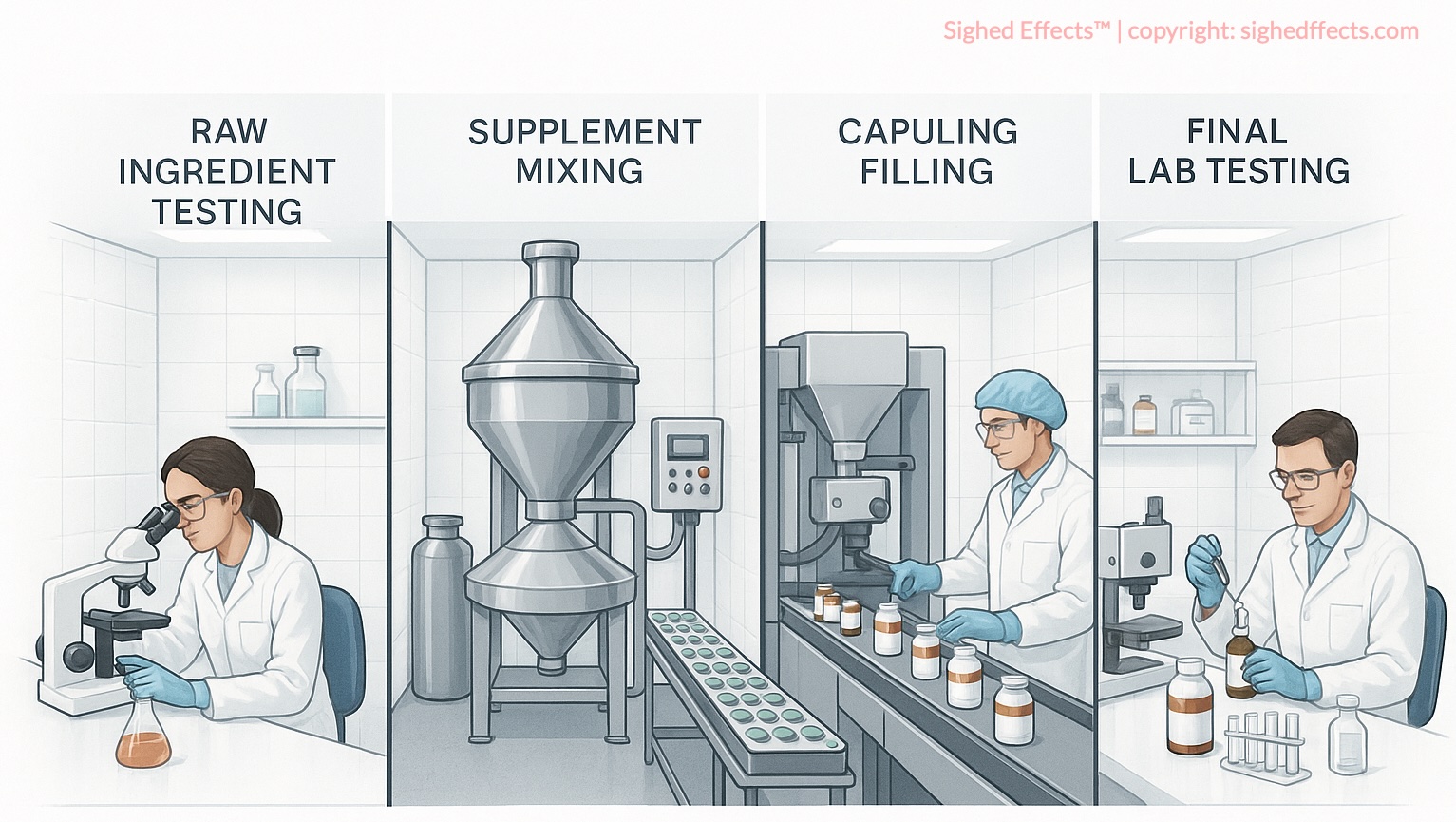
What in-house labs do during production
In-house quality control typically operates at several points in the manufacturing process. Understanding where these checkpoints fall helps clarify what in-house testing can and can’t catch.
The first checkpoint is raw material receipt. When ingredients arrive at the facility, the manufacturer is required under cGMP to verify their identity, confirming that what the supplier shipped is actually what was ordered. This is usually done with relatively quick analytical methods: Fourier-transform infrared spectroscopy (FTIR) for chemical fingerprinting, or simple colorimetric tests that match a sample against a known reference standard. These methods confirm that a shipment of magnesium glycinate is in fact magnesium glycinate and not magnesium oxide, but they typically don’t quantify purity or detect low-level contaminants.
During formulation and blending, in-house labs may run in-process checks, measuring the concentration of active ingredients as they’re mixed, confirming that the blend is homogeneous, and checking physical properties like moisture content. These checks are about manufacturing consistency: making sure the process is running correctly, not evaluating the final product as a consumer would receive it.
The final checkpoint is batch release testing on the finished product. This is where the manufacturer confirms that capsules, tablets, or powders meet internal specifications for potency (do the active ingredients match the target concentration?), basic microbial safety (are bacteria, yeast, and mold counts within acceptable limits?), and physical characteristics (do capsules seal properly, do tablets hold together?). Some companies also run disintegration tests to confirm that dosage forms will break down in the digestive tract.
The instruments involved vary by company budget and scale. Larger manufacturers may have HPLC systems for quantitative potency analysis and plating equipment for microbial counts. Smaller operations may rely on UV-visible spectrophotometry, which is cheaper but less precise, or outsource specific tests to contract labs that may or may not be independent of the brand.
Where the gaps are
The structural limitations of in-house testing aren’t about dishonesty. They’re about what equipment is available, what the testing protocol covers, and what falls outside both.
Most in-house labs don’t test for heavy metals. ICP-MS (the standard method for detecting lead, arsenic, cadmium, and mercury at parts-per-billion levels) is expensive equipment that requires trained operators and ongoing calibration. Many supplement manufacturers don’t have it. They may rely on supplier certificates of analysis for raw material purity rather than running their own contaminant screening on finished products. This means a contamination problem introduced during manufacturing, cross-contamination from shared equipment, environmental exposure during processing, could go undetected.
Adulteration and ingredient substitution are another blind spot. In-house identity testing confirms that an ingredient matches a known standard. It’s less effective at detecting sophisticated fraud: cheaper botanical species substituted for premium ones, synthetic compounds passed off as natural extracts, or amino acids added to protein powders to inflate nitrogen content (a practice known as amino spiking). Catching these requires more advanced analytical methods, DNA barcoding for botanicals, mass spectrometry for synthetic compound identification, that most internal labs aren’t equipped to run.
Batch consistency is a subtler issue. In-house testing typically analyzes a sample from each production run. But supplement manufacturing involves natural variability, especially with botanical ingredients where active compound concentrations differ between harvests, growing regions, and extraction conditions. A sample that tests well doesn’t guarantee the entire batch is uniform, and in-house labs rarely have the throughput to run the kind of statistical sampling that would catch within-batch variation.
The conflict of interest is real but often overstated in discussions of in-house testing. The more practical concern is selective testing scope: companies test for what they’re set up to test for, which tends to be potency of the headline ingredients. Contaminant screening, excipient verification, dissolution performance, and oxidation testing (for oil-based products) often fall outside the routine in-house protocol, not because anyone is hiding problems, but because the equipment and expertise required to run those panels aren’t available internally.

How to evaluate brands that reference in-house testing
When a supplement label says “lab tested” or “every batch tested,” the claim by itself tells you nothing about whether that testing was internal or independent, what was tested, or what the results were. These phrases are unregulated and can mean almost anything.
A brand that references its internal QC program credibly will typically be specific about what that program includes. The useful signals are whether they name the analytical methods they use, whether they test the finished product (not just raw materials), whether they publish batch-specific data linked to lot numbers, and whether they distinguish between their in-house testing and any independent third-party verification they’ve obtained. A company that clearly says “we test potency and microbial safety in-house, and we send finished products to [named ISO 17025 lab] for contaminant screening” is communicating honestly about a two-layer system.
The red flags are vagueness and conflation. “FDA-compliant lab” sounds authoritative but means little, the FDA doesn’t certify or approve supplement testing labs. “Pharmaceutical-grade testing” has no legal definition in the supplement context. Imagery of lab coats and chromatography equipment creates an impression of rigor without documenting what was actually tested. And internally generated Certificates of Analysis (COAs) that carry the brand’s own logo but don’t name an external laboratory are performing a different function than a COA from an accredited independent lab.
The most important question to ask is whether the brand’s in-house testing is supplemented by independent verification. In-house QC is a baseline. It’s what the law requires and what responsible manufacturing demands. But it operates within the company’s own system, tested by the company’s own people, on samples the company selected. For claims about contaminant-free status, label accuracy, or ingredient purity to carry weight with consumers who care about verification, those claims need outside confirmation. Our independent lab testing guide covers what that looks like and how to evaluate it.

In-house testing in context
In-house lab testing is a normal, necessary part of supplement manufacturing. It catches production errors, confirms ingredient identity, verifies potency of active compounds, and supports regulatory compliance. Companies that invest in capable internal labs and run thorough QC programs are doing more than the minimum, and that matters.
What in-house testing can’t provide is the external verification that closes the gap between manufacturer claims and consumer confidence. The equipment limitations, the scope constraints, and the inherent fact that the brand controls both the product and the data mean that in-house results, however well-intentioned, carry less evidentiary weight than results from an independent laboratory with no stake in the outcome.
The two aren’t alternatives, they’re layers. In-house testing handles the day-to-day quality control that keeps manufacturing on track. Independent testing provides the external check that confirms the system is working as claimed. For consumers evaluating a supplement brand’s credibility, the question isn’t whether the company does internal testing (most do, because they’re required to). The question is whether they’ve also submitted their product to someone outside the company who measured what’s in it and published what they found.

References
- 1
This article is part of our Certifications hub: Our deep dives into third-party testing, purity standards, and label verification systems across the supplement industry.