Vitamin E: 8 forms, skin claims, cancer risk, and what the research says
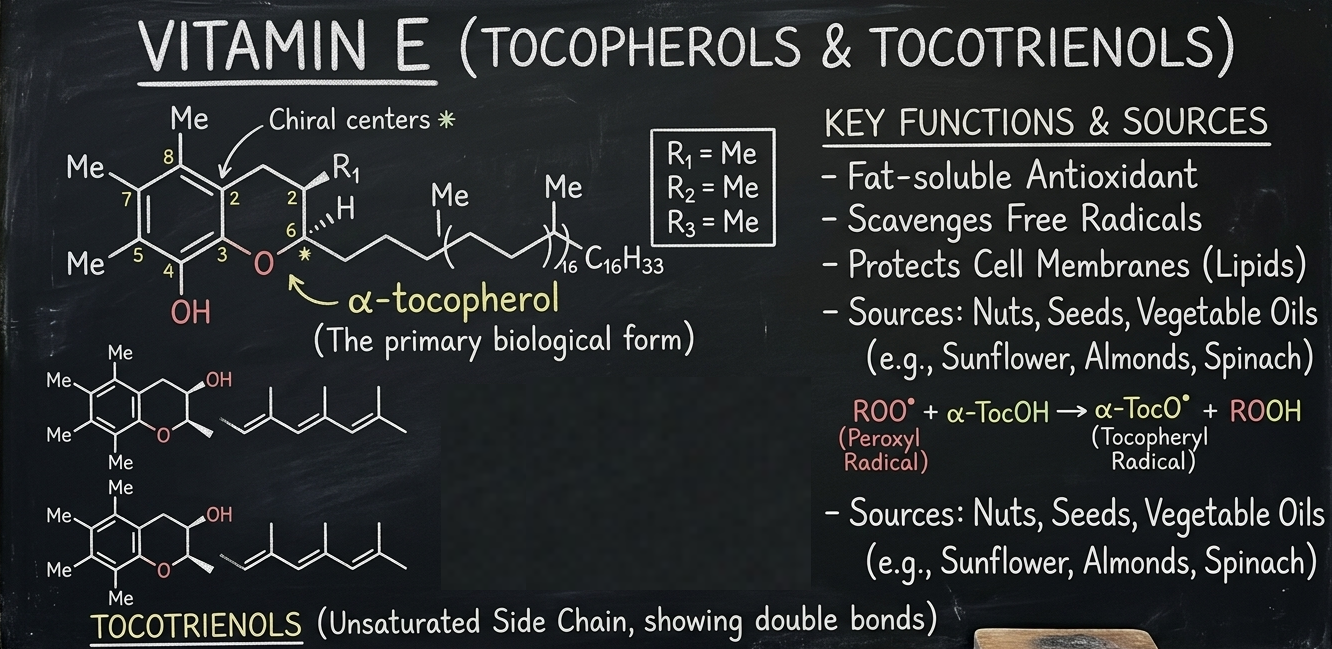
Vitamin E is one of those nutrients that sounds simple until you look at it closely. Most people think of it as a single thing — a vitamin you find in supplements and skin creams. In reality, “vitamin E” refers to a family of eight structurally related compounds, and the biology of how your body handles them is more selective and more interesting than the marketing suggests.
If you’ve been diagnosed with a fat malabsorption disorder or a condition affecting vitamin E transport, work with a medical provider. This article explains mechanisms and evidence. It is not a substitute for clinical evaluation.
What vitamin E actually is
The vitamin E family includes four tocopherols (alpha, beta, gamma, delta) and four tocotrienols (alpha, beta, gamma, delta). All eight share a basic architecture: a chromanol ring with a hydroxyl group that can donate hydrogen atoms to neutralize free radicals, attached to a long lipophilic tail that anchors the molecule into cell membranes. The difference between tocopherols and tocotrienols comes down to that tail — tocopherols have a fully saturated side chain, while tocotrienols have three double bonds in theirs, which makes them slightly more mobile within membranes.1Sen CK, Khanna S, Roy S. Tocotrienols: Vitamin E beyond tocopherols. Life Sci. 2006;78(18):2088-2098.
The distinction between the four forms within each class (alpha, beta, gamma, delta) depends on how many methyl groups sit on the chromanol ring and where they’re positioned. Alpha has the most methyl groups (three), delta has the fewest (one). This matters because the number and position of those methyl groups affect how tightly each form binds to the proteins your body uses to transport and retain vitamin E.
The biology gets selective at the liver. Your liver contains a protein called alpha-tocopherol transfer protein (α-TTP), and it has a strong preference for one specific form: RRR-alpha-tocopherol, the naturally occurring stereoisomer.2Institute of Medicine. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. Washington, DC: National Academies Press; 2000. When dietary fats carrying various vitamin E forms arrive at the liver via chylomicrons, α-TTP selectively binds alpha-tocopherol and packages it into lipoproteins for redistribution to tissues throughout the body. The other forms — gamma-tocopherol, delta-tocopherol, and all the tocotrienols — get metabolized and excreted relatively quickly.3Jiang Q. Metabolism of natural forms of vitamin E and biological actions of vitamin E metabolites. Free Radic Biol Med. 2022;179:375-395.
This is why the Food and Nutrition Board at the National Academy of Medicine defines the vitamin E requirement exclusively in terms of alpha-tocopherol. The other seven forms are absorbed from food, but your body doesn’t maintain meaningful blood or tissue levels of them the way it does alpha-tocopherol. Gamma-tocopherol is actually the dominant form in the American diet (it’s the primary tocopherol in soybean, corn, and canola oils), but plasma concentrations of gamma-tocopherol are typically only about a tenth of alpha-tocopherol levels because the liver preferentially retains alpha and clears everything else.4Jiang Q, Christen S, Shigenaga MK, Ames BN. Gamma-tocopherol, the major form of vitamin E in the US diet, deserves more attention. Am J Clin Nutr. 2001;74(6):714-722.
How it works in the body
Alpha-tocopherol’s primary job is acting as a chain-breaking antioxidant in cell membranes and plasma lipoproteins. The mechanism is straightforward: when a lipid peroxyl radical forms (a normal byproduct of oxidative metabolism that can propagate chain reactions damaging cell membranes), alpha-tocopherol donates a hydrogen atom from its hydroxyl group to the radical, neutralizing it. The resulting tocopheroxyl radical is relatively stable and doesn’t propagate the chain. It can then be recycled back to its active form by other antioxidants, particularly vitamin C, which is one of the better-documented examples of antioxidant synergy in human biochemistry.5Traber MG, Stevens JF. Vitamins C and E: beneficial effects from a mechanistic perspective. Free Radic Biol Med. 2011;51(5):1000-1013.
This membrane-protective function is especially relevant in tissues exposed to high oxidative stress — the lungs, the retina, red blood cells, and the skin. Alpha-tocopherol is the most abundant lipid-soluble antioxidant in human skin, with higher concentrations in the epidermis than the dermis.6Thiele JJ, Hsieh SN, Ekanayake-Mudiyanselage S. Vitamin E: critical review of its current use in cosmetic and clinical dermatology. Dermatol Surg. 2005;31(7 Pt 2):805-813. Exposure to UV radiation at doses below what would cause visible sunburn can deplete stratum corneum alpha-tocopherol by roughly half, which is part of why photoprotection has been such a focus of vitamin E research.
Beyond antioxidant activity, alpha-tocopherol has documented roles in immune function. It influences T-cell signaling and appears to support cell-mediated immunity, particularly in older adults where immune function tends to decline.7Meydani SN, Leka LS, Fine BC, et al. Vitamin E and respiratory tract infections in elderly nursing home residents: a randomized controlled trial. JAMA. 2004;292(7):828-836. There’s also evidence that alpha-tocopherol inhibits platelet aggregation through effects on protein kinase C, and that it modulates gene expression related to inflammation — though these functions operate at higher concentrations than what’s needed for basic antioxidant protection.
Food sources and how much you need
The RDA for vitamin E is 15 mg per day of alpha-tocopherol for adults, set by the Institute of Medicine in 2000.8Institute of Medicine. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. Washington, DC: National Academies Press; 2000. That number was based on a somewhat unusual endpoint — the amount of alpha-tocopherol needed to prevent hydrogen peroxide-induced hemolysis of red blood cells in a test-tube assay correlated with clinical observations of deficiency. The FNB itself acknowledged the limitations of this approach and called for research into better biomarkers. Still, 15 mg/day remains the standard.
Most Americans don’t hit that number from diet alone. Average intake from food runs around 7–11 mg/day depending on the survey, though these figures likely undercount because dietary fat intake (which carries much of the vitamin E) is chronically underreported in nutritional surveys.9Office of Dietary Supplements. Vitamin E Fact Sheet for Health Professionals. National Institutes of Health. Updated March 2025. Despite this apparent shortfall, clinical vitamin E deficiency is extremely rare in healthy adults. Using the standard plasma cutoff of 12 μmol/L, less than 1% of the U.S. adult population qualifies as inadequate.
The best food sources of alpha-tocopherol are wheat germ oil (the single richest source per serving), sunflower seeds, almonds, sunflower oil, and hazelnuts. A single ounce of sunflower seeds provides about 7.4 mg — roughly half the RDA. Almonds deliver around 6.8 mg per ounce. Peanut butter, spinach, and broccoli contribute smaller amounts but add up over the course of a day. For anyone eating a diet that includes nuts, seeds, and reasonable amounts of plant-based oils, meeting the RDA without supplementation is entirely achievable.
Because vitamin E is fat-soluble, absorption depends on dietary fat. Taking a vitamin E supplement on an empty stomach means you’ll absorb less of it compared to taking it with a meal containing some fat — a consideration that applies equally to <a href=”/supplement-bioavailability”>bioavailability</a> of all fat-soluble vitamins.
One labeling detail worth understanding: supplements come in either natural (d-alpha-tocopherol, which is the RRR stereoisomer) or synthetic (dl-alpha-tocopherol, which is a mix of eight stereoisomers). The natural form has roughly twice the bioactivity of the synthetic form because your body only retains the 2R-stereoisomers. So 15 mg of natural alpha-tocopherol meets the RDA, but you’d need 30 mg of synthetic to get the same effective dose. Supplement labels don’t always make this distinction clear.
Deficiency: rare but real
True vitamin E deficiency almost never results from poor diet. It’s overwhelmingly a consequence of disorders that impair fat absorption — cystic fibrosis, Crohn’s disease, chronic pancreatitis, short bowel syndrome, cholestatic liver disease — or of rare genetic conditions that disrupt vitamin E transport.
The most dramatic genetic form is ataxia with vitamin E deficiency (AVED), caused by mutations in the gene encoding alpha-tocopherol transfer protein. Without functional α-TTP, the liver can’t retain and redistribute alpha-tocopherol, and plasma levels crash. The result is progressive spinocerebellar ataxia (loss of coordinated movement), peripheral neuropathy, muscle weakness, and retinal degeneration. It’s treatable with high-dose vitamin E supplementation — typically 800–1,200 mg/day — but the damage is irreversible if not caught early.10Cavalier L, Ouahchi K, Kayden HJ, et al. Ataxia with isolated vitamin E deficiency: heterogeneity of mutations and phenotypic variability in a large number of families. Am J Hum Genet. 1998;62(2):301-310.
Abetalipoproteinemia is another rare inherited condition where patients can’t form chylomicrons properly, leading to severe fat-soluble vitamin malabsorption. These patients may require enormous vitamin E doses — in the range of 100 mg/kg body weight — to prevent neurological damage.
In people without these conditions, the body stores vitamin E in adipose tissue efficiently enough that it would take an estimated four years on a completely vitamin E-free diet to fully deplete stores in a healthy adult.11Vitamin E in dermatology. Indian Dermatol Online J. 2016;7(4):311-315. This is a long runway compared to water-soluble vitamins, and it explains why overt deficiency from dietary inadequacy essentially doesn’t happen in otherwise healthy people.
The claims: what the evidence actually says
Cancer prevention (and the SELECT reversal)
The story of vitamin E and cancer is one of the more instructive cautionary tales in supplement research. Early observational studies found correlations between higher vitamin E intake and lower cancer risk. Biological plausibility existed — antioxidants should, in theory, protect DNA from oxidative damage. And in 1994, the Alpha-Tocopherol, Beta-Carotene (ATBC) Cancer Prevention Study found a secondary result showing 32% fewer prostate cancers and 41% fewer prostate cancer deaths in Finnish male smokers taking 50 mg/day of alpha-tocopherol.12Heinonen OP, Albanes D, Virtamo J, et al. Prostate cancer and supplementation with alpha-tocopherol and beta-carotene: incidence and mortality in a controlled trial. J Natl Cancer Inst. 1998;90(6):440-446.
That secondary finding launched the Selenium and Vitamin E Cancer Prevention Trial (SELECT), one of the largest cancer prevention trials ever conducted. SELECT enrolled over 35,000 men across 427 sites in the U.S., Canada, and Puerto Rico, randomized to receive selenium, vitamin E (400 IU/day of synthetic alpha-tocopheryl acetate), both, or placebo.13Klein EA, Thompson IM Jr, Tangen CM, et al. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA. 2011;306(14):1549-1556.
The trial was stopped early in 2008 because it was clear vitamin E was not preventing prostate cancer. When follow-up data were published in 2011, the result was worse than neutral: men taking vitamin E alone had a 17% increase in prostate cancer risk compared to placebo — 76 cases per 1,000 men versus 65 cases per 1,000 men over a seven-year period. That difference was statistically significant.14Klein EA, Thompson IM Jr, Tangen CM, et al. Vitamin E and the risk of prostate cancer: the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA. 2011;306(14):1549-1556.
A couple of things are worth noting about this result. First, SELECT used 400 IU/day of synthetic alpha-tocopherol — roughly 27 times the RDA and a dose that no one was getting from food. Second, a subsequent analysis looking at toenail selenium levels (a reliable biomarker of selenium status) found that the vitamin E risk was concentrated in men who started the trial with low selenium levels. Men with adequate selenium who took vitamin E did not show the same increase.15Kristal AR, Darke AK, Morris JS, et al. Baseline selenium status and effects of selenium and vitamin E supplementation on prostate cancer risk. J Natl Cancer Inst. 2014;106(3):djt456. This hints at a nutrient interaction — possibly that selenium is needed to buffer some effect of high-dose alpha-tocopherol — but the mechanism remains unclear.
The broader point is that SELECT did exactly what a well-designed trial is supposed to do: test a hypothesis generated by observational data under controlled conditions. The hypothesis failed. High-dose synthetic alpha-tocopherol supplementation does not prevent prostate cancer and may increase risk.
The high-dose mortality question
In 2005, a meta-analysis by Miller et al. pooled data from 19 randomized trials (involving over 135,000 participants) and found that vitamin E supplementation at doses of 400 IU/day or higher was associated with a small but statistically significant increase in all-cause mortality — about 39 additional deaths per 10,000 persons compared to controls.16Miller ER 3rd, Pastor-Barriuso R, Dalal D, Riemersma RA, Appel LJ, Guallar E. Meta-analysis: high-dosage vitamin E supplementation may increase all-cause mortality. Ann Intern Med. 2005;142(1):37-46.
This finding received enormous media attention, but it comes with caveats. The high-dose trials included in the meta-analysis were small and overwhelmingly enrolled people with existing chronic diseases — heart disease, cancer, renal failure, Alzheimer’s. Whether the same risk applies to healthy adults taking moderate doses remains genuinely unclear. A later meta-analysis by Abner et al. (2011) that included additional trials and broader populations found no relationship between vitamin E dose and mortality risk, suggesting the original result may have been driven by the specific mix of trials included.17Abner EL, Schmitt FA, Mendiondo MS, Marcum JL, Kryscio RJ. Vitamin E and all-cause mortality: a meta-analysis. Curr Aging Sci. 2011;4(2):158-170.
One proposed mechanism for harm at high doses is that large amounts of alpha-tocopherol may displace other fat-soluble antioxidants — gamma-tocopherol in particular — from cell membranes and lipoproteins. Since gamma-tocopherol has some unique properties (it can trap reactive nitrogen species that alpha-tocopherol can’t), flooding the system with synthetic alpha may actually reduce total antioxidant coverage. There’s also the possibility that very high-dose alpha-tocopherol acts as a pro-oxidant under certain cellular conditions, though this is better established in vitro than in vivo.
The practical takeaway: supplementation at or below the RDA (15 mg/day) has not been associated with harm. Supplementation in the 200–400 IU/day range used in cardiovascular and cancer trials has shown no benefit and possible risk. There is no good evidence supporting megadose vitamin E supplementation for any purpose in healthy adults.
Skin, aging, and the scar myth
Vitamin E is a massive ingredient in the cosmetics industry. Anti-aging creams and scar treatments rely on it, and it’s a staple of post-surgical wound care ointments — most contain 0.5%–1% vitamin E, and it’s one of the top reasons consumers buy vitamin E capsules to break open and apply directly to skin.
The premise isn’t unreasonable. Alpha-tocopherol is the predominant lipid-soluble antioxidant in the stratum corneum (the outermost skin layer), and UV exposure depletes it rapidly. In animal models, topical alpha-tocopherol applied before UV exposure reduces sunburn cells, DNA damage, and erythema. Human studies have shown similar photoprotective effects when vitamin E is applied topically before sun exposure, particularly in combination with vitamin C.18Lin JY, Selim MA, Shea CR, et al. UV photoprotection by combination topical antioxidants vitamin C and vitamin E. J Am Acad Dermatol. 2003;48(6):866-874.
The problem is that photoprotection before UV exposure and treatment of existing skin damage are different things. Despite decades of use, there is very little controlled clinical evidence that topical vitamin E improves the cosmetic appearance of scars, reverses photoaging, or heals existing skin damage.
The most-cited study on this question is a 1999 double-blind trial by Baumann and Spencer. Fifteen patients who had undergone skin cancer surgery applied either an emollient containing vitamin E or the emollient alone to their scars twice daily for four weeks. The result: no improvement in scar appearance from vitamin E, and 33% of patients developed contact dermatitis from the vitamin E preparation.19Baumann LS, Spencer J. The effects of topical vitamin E on the cosmetic appearance of scars. Dermatol Surg. 1999;25(4):311-315. A 2016 systematic review of all prospective studies on topical vitamin E for scar management found only six studies that met inclusion criteria. Of those, the three that used vitamin E as monotherapy showed no significant benefit.20Tanaydin V, Conings J, Malyar M, van der Hulst R, van der Lei B. The role of topical vitamin E in scar management: a systematic review. Aesthetic Surg J. 2016;36(8):959-965.
The skincare industry isn’t lying exactly — vitamin E does protect cell membranes from oxidative damage, and keeping skin moisturized does improve its appearance. But the active ingredient in most vitamin E skin products is probably the oil base doing basic emollient work, not the vitamin E performing some special anti-aging function. The gap between “vitamin E is the dominant antioxidant in skin” and “rubbing vitamin E on skin reverses aging or heals scars” is enormous, and the clinical evidence does not bridge it.
There’s also an important distinction between forms. Free (unesterified) alpha-tocopherol can donate hydrogen atoms and function as an antioxidant directly. But many topical products use alpha-tocopheryl acetate — an ester conjugate that’s more stable on the shelf. The problem is that skin has limited ability to convert tocopheryl acetate back to free tocopherol, so the antioxidant benefit of esterified preparations applied topically may be minimal.21Thiele JJ, Ekanayake-Mudiyanselage S. Vitamin E in human skin: organ-specific physiology and considerations for its use in dermatology. Mol Aspects Med. 2007;28(5-6):646-667.
Blood thinning and anticoagulant interactions
Vitamin E gets flagged as a blood thinner in many drug interaction databases, and people on warfarin are frequently warned about it. The mechanism is real but the clinical magnitude is often overstated.
Alpha-tocopherol inhibits platelet aggregation through effects on protein kinase C signaling and thromboxane A2 synthesis. At high doses, it can also interfere with vitamin K-dependent carboxylation of clotting factors — essentially competing with vitamin K in the coagulation cascade. In animal models where vitamin K was depleted by warfarin, additional high-dose vitamin E further reduced clotting factor activity.22Corrigan JJ Jr, Ulfers LL. Effect of vitamin E on prothrombin levels in warfarin-induced vitamin K deficiency. Am J Clin Nutr. 1981;33(7):1594-1600.
But in the best controlled human study on this interaction — a double-blind trial randomizing 21 patients on chronic warfarin therapy to receive either vitamin E (800–1,200 IU/day) or placebo — none of the patients receiving vitamin E had a significant change in their INR (the standard measure of anticoagulation).23Kim JM, White RH. Effect of vitamin E on the anticoagulant response to warfarin. Am J Cardiol. 1996;77(7):545-546. The authors concluded that moderate to large doses of vitamin E could safely be used in patients receiving warfarin, though they recommended checking INR after starting supplementation as a routine precaution.
A comprehensive 2014 review of vitamin E-drug interactions concluded that there is no evidence from animal models or randomized human trials suggesting that vitamin E at nutritionally relevant doses (below 300 mg/day) causes adverse drug interactions. The interaction concern is dose-dependent and most relevant above 300–400 mg/day, particularly in people who are already vitamin K deficient or on anticoagulant therapy.24Podszun M, Frank J. Vitamin E-drug interactions: molecular basis and clinical relevance. Nutr Res Rev. 2014 Dec;27(2):215-31.
The practical guidance: if you take warfarin or another vitamin K antagonist and want to supplement with vitamin E, keep the dose modest (under 200–400 IU/day) and have your INR checked within a couple weeks of starting. If you’re not on anticoagulants and you’re taking a normal multivitamin dose, this isn’t something you need to worry about.
Heart disease prevention
The vitamin E and heart disease story follows a depressingly familiar arc. Observational data from the Nurses’ Health Study and the Health Professionals Follow-Up Study in the early 1990s found that people taking vitamin E supplements had lower rates of coronary disease. The mechanism was plausible: alpha-tocopherol prevents oxidation of LDL cholesterol, and oxidized LDL is a driver of atherosclerotic plaque formation.
Then the randomized controlled trials came in. The HOPE trial (2000), the GISSI-Prevenzione trial (1999), the Heart Protection Study (2002), the Women’s Health Study (2005) — large, well-powered, well-conducted trials involving tens of thousands of participants collectively — all found no cardiovascular benefit from vitamin E supplementation.25Vivekananthan DP, Penn MS, Sapp SK, Hsu A, Topol EJ. Use of antioxidant vitamins for the prevention of cardiovascular disease: meta-analysis of randomised trials. Lancet. 2003;361(9374):2017-2023. The hypothesis that you could pop an antioxidant pill and prevent heart disease turned out to be wrong, at least with alpha-tocopherol at the doses tested.
One possible exception involves people with type 2 diabetes who carry the haptoglobin 2-2 genotype, a variant that impairs the body’s ability to clear free hemoglobin (a potent oxidant). A subset analysis of the HOPE trial and a subsequent study (ICARE) showed that vitamin E supplementation reduced cardiovascular events specifically in this genotype group.26Milman U, Blum S, Shapira C, et al. Vitamin E supplementation reduces cardiovascular events in a subgroup of middle-aged individuals with both type 2 diabetes mellitus and the haptoglobin 2-2 genotype. Arterioscler Thromb Vasc Biol. 2008;28(2):341-347. This is a niche finding — roughly 40% of diabetics carry haptoglobin 2-2 — but it illustrates a recurring theme in vitamin E research: blanket supplementation in healthy populations doesn’t work, but targeted use in specific contexts with identified oxidative stress might.
The tocopherol vs. mixed tocopherols debate
Most vitamin E supplements contain only alpha-tocopherol. Some researchers and supplement manufacturers argue this is a mistake — that supplementing with alpha-tocopherol alone displaces gamma-tocopherol and other forms, potentially doing more harm than good, and that mixed-tocopherol or mixed-tocotrienol formulas are superior.
There’s a kernel of truth here. High-dose alpha-tocopherol supplementation does reduce plasma gamma-tocopherol concentrations, and gamma-tocopherol has properties that alpha-tocopherol doesn’t — it can trap reactive nitrogen species and has anti-inflammatory effects through cyclooxygenase-2 inhibition that appear independent of antioxidant activity. In cell culture studies, tocotrienols show stronger anti-proliferative effects against cancer cell lines than tocopherols, and at lower concentrations.
But the clinical translation of these findings has been limited. Human trials using tocotrienol supplements have been described by researchers in the field as “pretty disappointing,” primarily because tocotrienols have poor bioavailability — they compete with tocopherols for the same carrier proteins in the gut, and the liver’s alpha-TTP preferentially clears them from circulation.27Sylvester PW. The other vitamin E. AOCS Lipid Library. 2013. Taking tocotrienols along with tocopherols actually reduces tocotrienol absorption, creating a kind of catch-22 for mixed-vitamin E formulas.
The idea that a complete “full-spectrum” vitamin E supplement is inherently better than alpha-tocopherol alone is popular in supplement marketing but is not well supported by human outcomes data. The forms have different biological properties in laboratory settings; whether those differences translate into meaningfully different health effects in people taking oral supplements remains an open question.
Who might actually benefit from supplementation
Given the largely disappointing trial data, who has a reasonable evidence-based case for vitamin E supplementation?
People with documented fat malabsorption disorders — cystic fibrosis, Crohn’s disease, short bowel syndrome, chronic cholestatic liver disease — are at genuine risk of deficiency and may benefit from supplementation under medical supervision. This is straightforward clinical nutrition, not speculative supplementation.
There is some evidence supporting vitamin E supplementation in nonalcoholic steatohepatitis (NASH), the inflammatory form of fatty liver disease. The PIVENS trial found that 800 IU/day of vitamin E improved liver histology in non-diabetic adults with NASH, and several hepatology guidelines now include vitamin E as a treatment option for specific NASH populations.28Sanyal AJ, Chalasani N, Kowdley KV, et al. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N Engl J Med. 2010;362(18):1675-1688.
The AREDS2 formula for age-related macular degeneration includes vitamin E (400 IU) as part of a combination with vitamin C, zinc, copper, and lutein/zeaxanthin. The evidence supports the combination, not vitamin E alone, for slowing progression of intermediate to advanced AMD.
For healthy adults with adequate dietary intake, the evidence does not support routine vitamin E supplementation for cancer or cardiovascular disease prevention. The data on cognitive decline is similarly unconvincing. The American Heart Association, the U.S. Preventive Services Task Force, and the National Cancer Institute have all issued statements to this effect.
Dosing and safety
The tolerable upper intake level (UL) for vitamin E is 1,000 mg/day (roughly 1,500 IU of natural alpha-tocopherol or 1,100 IU of synthetic). This ceiling was set based on the risk of hemorrhagic effects — at very high doses, vitamin E’s antiplatelet activity can increase bleeding risk.29Institute of Medicine. Dietary Reference Intakes for Vitamin C, Vitamin E, Selenium, and Carotenoids. Washington, DC: National Academies Press; 2000.
For general supplementation (if you choose to supplement at all), staying in the range of 15–200 mg/day of natural alpha-tocopherol is reasonable and has not been associated with harm in controlled trials. Doses in the 400 IU+ range used in large clinical trials have not shown benefit for any condition in healthy populations and carry uncertain long-term risk.
If you’re buying a vitamin E supplement, look for d-alpha-tocopherol (the natural form) rather than dl-alpha-tocopherol (synthetic). The natural form is better retained by the body. Whether mixed tocopherol formulas offer meaningful advantages over alpha-tocopherol alone is not settled by the available human data.
Vitamin E should be taken with a meal containing some fat for optimal absorption. If you take anticoagulant medications, discuss vitamin E supplementation with your prescriber and monitor your coagulation status after starting.
If you’re looking for help figuring out whether vitamin E fits into your supplement routine and at what dose, the supplements overview and bioavailability guide on this site are good starting points for understanding how fat-soluble vitamins work in context.
The pattern with vitamin E
Vitamin E’s trajectory in nutrition research follows a pattern that repeats across supplement science: observational data suggests a benefit, biological plausibility exists, and then randomized controlled trials either fail to confirm the benefit or reveal harm. This happened with beta-carotene and lung cancer. It happened with vitamin A supplementation. And it happened with vitamin E and both prostate cancer and cardiovascular disease.
The lesson isn’t that vitamin E is useless — it’s an essential nutrient with well-characterized functions in membrane protection and immune regulation. But there’s a meaningful difference between correcting a deficiency and megadosing a nutrient in hopes of pharmacological benefit. Your body’s system for handling vitamin E is precisely calibrated: a specific transfer protein selects a specific form, maintains specific plasma levels, and discards the rest. Flooding that system with 400 IU/day of synthetic alpha-tocopheryl acetate is not giving the system more of what it wants. It’s overriding the regulatory architecture.
For most people, the best approach to vitamin E is a diet that includes nuts, seeds, and plant-based oils — the foods your body evolved to extract this nutrient from. If you have a specific clinical condition where evidence supports supplementation, work with a provider to determine the appropriate form and dose. And if you’re rubbing vitamin E oil on a scar because someone told you it would help, the evidence says you’re mostly paying for expensive moisturizer.
Generate Your Stack. Avoid Conflicts. Optimize Absorption.
Most supplement advice ignores timing, interactions, and dose-response curves. Smart Stack Builder doesn't. Get a personalized daily schedule with interaction flags and dosage guidance, built on the same evidence base as this article.
References
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
- 15
- 16
- 17
- 18
- 19
- 20
- 21
- 22
- 23
- 24
- 25
- 26
- 27
- 28
- 29
This article is part of our Supplements hub: A science-backed collection of ingredient research, clinical dosage reviews, and optimized stack insights.