Folate (vitamin B9): the folate cycle, folic acid vs. methylfolate, and neural tube defect prevention
Folate
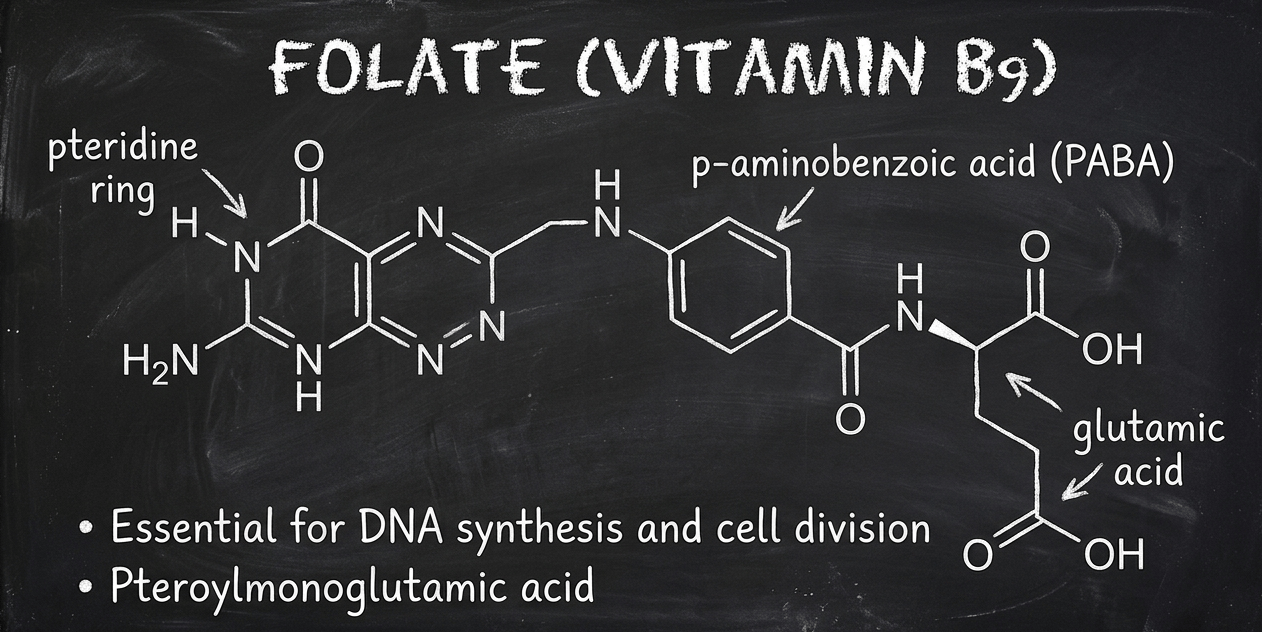
Folate is vitamin B9, but calling it a single vitamin undersells the complexity. The term covers a family of compounds built around a pteridine ring linked to para-aminobenzoic acid and one or more glutamate residues. Food folates exist as polyglutamate forms (multiple glutamates attached). Folic acid, the synthetic form used in supplements and food fortification, is a monoglutamate with the pteridine ring in its fully oxidized state. 5-methyltetrahydrofolate (5-MTHF), often sold as methylfolate, is the predominant circulating form in blood and the form most commonly used in newer supplements. These are not interchangeable molecules in the body, and the distinctions between them matter for absorption and metabolism, with downstream effects on clinical outcomes.
What unifies all folate forms is their function: carrying and donating one-carbon units. The folate cycle is the central switching station for one-carbon metabolism, the biochemical network that supplies methyl groups for DNA synthesis, epigenetic regulation, amino acid recycling, and neurotransmitter production. Without adequate folate, cells cannot replicate their DNA, methionine cannot be regenerated from homocysteine, and the methylation reactions that regulate gene expression stall.
This article covers the folate cycle in mechanistic detail, the different supplemental forms and what the evidence says about each, the neural tube defect prevention story (one of the strongest supplement recommendations in all of nutrition), the MTHFR polymorphism, and the safety concerns that emerge at high intakes. The B vitamins overview covers folate’s relationship to the other B vitamins at a systems level; this article goes deeper into folate’s own biochemistry.
A necessary disclaimer: If you are pregnant, planning to become pregnant, or taking medications that interfere with folate metabolism (including methotrexate, certain anticonvulsants, or sulfasalazine), work with a qualified medical professional. Folate status during early pregnancy has direct consequences for fetal development. This article is educational. It is not a treatment plan, a diagnostic tool, or a substitute for clinical care.
The folate cycle
The folate cycle is best understood as a system for loading, carrying, and unloading one-carbon units at different oxidation states. Tetrahydrofolate (THF) is the unloaded carrier. When THF picks up a one-carbon unit, it becomes a substituted folate, and the identity of that substituted form determines which biosynthetic reaction it can participate in.
Loading the carrier
The primary source of one-carbon units is serine. Serine hydroxymethyltransferase (SHMT), a PLP-dependent enzyme, cleaves serine into glycine and transfers the hydroxymethyl group to THF, producing 5,10-methylenetetrahydrofolate (5,10-methylene-THF).1Stover, P.J. and Field, M.S. “Trafficking of Intracellular Folates.” Advances in Nutrition, 2011; 2(4): 325-331. This is the most metabolically versatile folate intermediate. From 5,10-methylene-THF, the one-carbon unit can proceed in one of two directions: it can be used directly for nucleotide synthesis, or it can be further reduced to 5-methyltetrahydrofolate (5-MTHF) for homocysteine remethylation.
Histidine, glycine, and formate also contribute one-carbon units, but serine is quantitatively the most important source. The mitochondria play a significant but often overlooked role here: mitochondrial one-carbon metabolism generates formate from serine, which is exported to the cytoplasm and incorporated into the folate pool as 10-formyl-THF.2Tibbetts, A.S. and Appling, D.R. “Compartmentalization of Mammalian Folate-Mediated One-Carbon Metabolism.” Annual Review of Nutrition, 2010; 30: 57-81.
Nucleotide synthesis: the thymidylate branch
5,10-methylene-THF is the one-carbon donor for thymidylate synthase, the enzyme that converts deoxyuridine monophosphate (dUMP) to deoxythymidine monophosphate (dTMP). This is the only de novo pathway for producing thymidine, one of the four nucleotide bases in DNA. When this reaction occurs, the folate cofactor is oxidized to dihydrofolate (DHF), which must be recycled back to THF by dihydrofolate reductase (DHFR) before it can accept another one-carbon unit.3Fox, J.T. and Stover, P.J. “Folate-Mediated One-Carbon Metabolism.” Vitamins and Hormones, 2008; 79: 1-44.
This thymidylate synthesis pathway is why folate deficiency impairs DNA replication. Without enough 5,10-methylene-THF, cells cannot produce thymidine at the rate needed for DNA synthesis. Uracil gets misincorporated into DNA in place of thymidine, triggering futile repair cycles that fragment chromosomes. Rapidly dividing cells are affected first, which is why folate deficiency manifests as megaloblastic anemia (the bone marrow cannot produce normal red blood cells) and why folate is so critical during embryonic development, when cell division is at its most intense.
A separate branch of 10-formyl-THF supplies two of the carbon atoms (C-2 and C-8) in the purine ring during de novo purine synthesis. Folate deficiency thus impairs both pyrimidine (thymidylate) and purine biosynthesis, affecting the entire nucleotide pool.
Homocysteine remethylation: the methylation branch
The other fate of 5,10-methylene-THF is reduction to 5-methyltetrahydrofolate (5-MTHF) by the enzyme methylenetetrahydrofolate reductase (MTHFR). This reaction is essentially irreversible under physiological conditions and uses riboflavin (as FAD) as a cofactor.4Froese, D.S., Fowler, B., and Baumgartner, M.R. “Vitamin B12, Folate, and the Methionine Remethylation Cycle—Biochemistry, Pathways, and Regulation.” Journal of Inherited Metabolic Disease, 2019; 42(4): 673-685.
5-MTHF is the predominant folate form in blood and the methyl group donor for methionine synthase, the vitamin B12-dependent enzyme that converts homocysteine to methionine. In this reaction, the methyl group passes from 5-MTHF to cobalamin (forming methylcobalamin), then from methylcobalamin to homocysteine, regenerating both methionine and THF. The regenerated THF re-enters the folate cycle to accept another one-carbon unit, completing the loop.
This is the only reaction that can release the methyl group from 5-MTHF. If methionine synthase is impaired (because of B12 deficiency, for example), 5-MTHF accumulates and THF cannot be regenerated, trapping cellular folate in a metabolically useless form. This is the “methyl trap” described in the B12 article and the B vitamins overview: B12 deficiency creates a functional folate deficiency by locking folate in the 5-MTHF form, starving the thymidylate synthesis pathway of 5,10-methylene-THF even when total folate is adequate.
Why the cycle matters
The folate cycle sits at the intersection of DNA integrity and cellular methylation. The two branches compete for the same substrate: 5,10-methylene-THF can go toward thymidylate synthesis (DNA) or toward 5-MTHF production (methylation). MTHFR activity determines how much folate is committed to the methylation pathway versus kept available for nucleotide synthesis. This partitioning explains why folate status affects both cancer biology (through DNA synthesis and repair) and cardiovascular risk (through homocysteine metabolism), and why the MTHFR polymorphism has attracted so much clinical attention.
The methionine generated by the methylation branch is converted to S-adenosylmethionine (SAM), the body’s primary methyl donor. SAM provides methyl groups for DNA methylation (epigenetic gene regulation), histone methylation (chromatin structure), neurotransmitter synthesis and degradation, phospholipid metabolism, and creatine synthesis, among others. Adequate folate is therefore a prerequisite for the entire methylation economy of the cell, not just for DNA synthesis.
Absorption and forms
Food folates are polyglutamate forms that must be hydrolyzed to monoglutamates by the brush border enzyme glutamate carboxypeptidase II (also called folylpoly-γ-glutamate carboxypeptidase) before absorption in the proximal small intestine. This hydrolysis step is a rate-limiting factor: food folate bioavailability averages roughly 50% of that of synthetic folic acid, which is already a monoglutamate and does not require this processing.5Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: National Academies Press, 1998. To account for this difference, folate requirements are expressed in dietary folate equivalents (DFE): 1 μg DFE = 1 μg food folate = 0.6 μg folic acid taken with food = 0.5 μg folic acid taken on an empty stomach.
Once absorbed, folate must be converted to its active coenzyme forms. The processing pathway differs by form:
Food folate (polyglutamate THF derivatives) is hydrolyzed to monoglutamate, absorbed, and enters the folate pool primarily as 5-MTHF after first-pass metabolism in the intestinal mucosa and liver. This is the natural entry point and does not require DHFR.
Folic acid (pteroylglutamic acid) is synthetic and fully oxidized. It must be reduced first to dihydrofolate and then to tetrahydrofolate by dihydrofolate reductase (DHFR) before it can enter one-carbon metabolism. Human DHFR activity is relatively slow compared to that in rodents, and at oral doses above approximately 200-300 μg, the enzyme begins to saturate.6Bailey, S.W. and Ayling, J.E. “The extremely slow and variable activity of dihydrofolate reductase in human liver and its implications for high folic acid intake.” Proceedings of the National Academy of Sciences, 2009; 106(36): 15424-15429. Above this threshold, unmetabolized folic acid (UMFA) appears in the bloodstream. The biological significance of circulating UMFA is debated, but its presence indicates that the conversion capacity has been exceeded.
5-MTHF (methylfolate) supplements bypass the DHFR step entirely, entering the folate pool in its already-reduced form. This is the primary selling point: 5-MTHF does not depend on DHFR capacity and does not generate unmetabolized folic acid. For individuals with reduced MTHFR activity (discussed below), 5-MTHF supplementation provides the end product directly.
One important nuance: folic acid produces similar plasma and red blood cell folate concentrations to equimolar doses of 5-MTHF in clinical studies, regardless of MTHFR genotype.7Prinz-Langenohl, R., Brämswig, S., Tobolski, O., Smulders, Y.M., Smith, D.E.C., Finglas, P.M., and Pietrzik, K. “6[S]-5-methyltetrahydrofolate increases plasma folate more effectively than folic acid in women with the homozygous or wild-type 677C→T polymorphism of methylenetetrahydrofolate reductase.” British Journal of Pharmacology, 2009; 158(8): 2014-2021. The CDC states that folic acid intake is more important for determining blood folate levels than having an MTHFR variant. The practical difference between the two forms is most relevant at high doses (where DHFR saturation and UMFA become factors) and in individuals with homozygous MTHFR C677T variants (where reduced MTHFR activity compounds the issue).
Neural tube defect prevention
Folate supplementation before and during early pregnancy to prevent neural tube defects (NTDs) is one of the best-established recommendations in nutritional science. The neural tube closes by day 28 of gestation, often before a woman knows she is pregnant, which is why supplementation must begin before conception.
The 1991 Medical Research Council Vitamin Study, a landmark randomized controlled trial, found that 4 mg/day of folic acid reduced the recurrence of NTDs by 72% in women with a prior NTD-affected pregnancy.8MRC Vitamin Study Research Group. “Prevention of neural tube defects: results of the Medical Research Council Vitamin Study.” Lancet, 1991; 338(8760): 131-137. Subsequent studies established that 400 μg/day of folic acid reduces the first occurrence of NTDs by a similar magnitude. The Berry et al. study in China found a 79% reduction in NTD risk with 400 μg/day folic acid supplementation among women with no prior NTD history.9Berry, R.J., Li, Z., Erickson, J.D., Li, S., Moore, C.A., Wang, H., Mulinare, J., Zhao, P., Wong, L.Y.C., Gindler, J., Hong, S.X., and Correa, A. “Prevention of neural-tube defects with folic acid in China.” New England Journal of Medicine, 1999; 341(20): 1485-1490.
These findings led the U.S. Public Health Service to recommend in 1992 that all women of childbearing age consume 400 μg of folic acid daily. Because roughly half of pregnancies are unplanned, and because supplement adherence in the target population was insufficient, the FDA mandated in 1998 that all enriched grain products in the United States be fortified with 140 μg of folic acid per 100 g of flour. More than 60 countries now have mandatory folic acid fortification programs.
The impact has been significant. NTD-affected pregnancies in the United States declined from approximately 4,000 per year before fortification to roughly 3,000 per year after. The reduction is estimated at 25-50% depending on the population and surveillance methodology.10Williams, J., Mai, C.T., Mulinare, J., Isenburg, J., Flood, T.J., Ethen, M., Frohnert, B., and Kirby, R.S. “Updated estimates of neural tube defects prevented by mandatory folic acid fortification — United States, 1995-2011.” Morbidity and Mortality Weekly Report, 2015; 64(1): 1-5. A prevalence floor of approximately 5-6 cases per 10,000 pregnancies appears to represent the limit of what folic acid can achieve, with the remaining NTDs likely being folate-insensitive.
The critical importance of this evidence cannot be overstated, and it has implications for the folic acid vs. 5-MTHF debate. Every randomized trial demonstrating NTD prevention used folic acid. There are currently no published clinical studies demonstrating that 5-MTHF supplementation prevents NTDs.11De-Regil, L.M., Peña-Rosas, J.P., Fernández-Gaxiola, A.C., and Rayco-Solon, P. “Effects and safety of periconceptional oral folate supplementation for preventing birth defects.” Cochrane Database of Systematic Reviews, 2015; 12: CD007950. The biological plausibility is there, since the mechanism of NTD prevention presumably involves the same folate-dependent pathways, but the clinical evidence base is entirely built on folic acid.
The MTHFR polymorphism
The MTHFR C677T polymorphism is the most common inborn error of folate metabolism. The variant produces a thermolabile enzyme with reduced activity: approximately 65% residual activity in heterozygotes (CT genotype) and approximately 30% residual activity in homozygotes (TT genotype). The TT genotype is found in 10-15% of North American and European populations and up to 25% in some Hispanic and Italian populations.12Frosst, P., Blom, H.J., Milos, R., Goyette, P., Sheppard, C.A., Matthews, R.G., Boers, G.J.H., den Heijer, M., Kluijtmans, L.A.J., van den Heuvel, L.P., and Rozen, R. “A candidate genetic risk factor for vascular disease: a common mutation in methylenetetrahydrofolate reductase.” Nature Genetics, 1995; 10(1): 111-113.
Reduced MTHFR activity means less 5,10-methylene-THF is converted to 5-MTHF. The consequences are a modest reduction in methionine regeneration, mildly elevated homocysteine (particularly when folate intake is low), and a shift in the folate pool toward 5,10-methylene-THF (which is actually the substrate for thymidylate synthesis). TT homozygotes have lower plasma folate and lower red blood cell folate than CC homozygotes, and they have modestly elevated homocysteine levels, but these effects are most pronounced when folate intake is inadequate and largely corrected by adequate folate intake from any source.
The CDC is explicit on this point: people with MTHFR gene variants can process all types of folate, including folic acid, and folic acid intake is more important for determining blood folate levels than MTHFR status. A large population-based trial in China found that 400 μg/day of folic acid effectively reduced NTD risk regardless of MTHFR genotype, and achieved target red blood cell folate concentrations across all genotype groups.
This does not mean the polymorphism is clinically irrelevant. TT homozygotes with low folate intake are at genuinely increased risk for hyperhomocysteinemia, and the epidemiological associations between the TT genotype and NTD risk, cardiovascular disease, and some cancers are real, particularly in folate-depleted populations. But the solution in most cases is adequate folate intake, not a specific form of folate. Whether 5-MTHF supplementation offers a meaningful clinical advantage over folic acid for TT homozygotes with adequate folate status remains an open question without a definitive trial answer. Riboflavin (B2) status is also relevant, since FAD is a cofactor for MTHFR, and riboflavin supplementation has been shown to lower homocysteine in TT homozygotes independently of folate.
The fortification tradeoff
Mandatory folic acid fortification is a public health success for NTD prevention, but it created a population-wide exposure to synthetic folic acid that was not without concerns.
The most clinically significant concern is the masking of <a href=”/vitamin-b12″>B12 deficiency</a>. Folic acid can correct the megaloblastic anemia caused by B12 deficiency (because it partially bypasses the methyl trap by providing substrate for the thymidylate pathway), which may normalize blood counts while B12-dependent neurological damage continues. This was the primary reason the IOM set the tolerable upper intake level (UL) for folic acid at 1,000 μg/day. In practice, modern clinical evaluation that includes MMA and homocysteine testing has reduced the likelihood of missed B12 deficiency diagnoses, but the concern remains valid, particularly in older adults who may not receive comprehensive workups.
The cancer question is more nuanced. Folate has a dual role in cancer biology that depends on timing and dose. In healthy tissue, adequate folate protects against cancer by supporting accurate DNA synthesis and repair. In cells that have already undergone malignant transformation, folate may promote tumor growth by supplying the nucleotides that rapidly dividing cancer cells need. Animal studies have demonstrated this dual effect, and some observational data in humans raised concerns about increased colorectal cancer incidence in the years following mandatory fortification.13Kim, Y.I. “Will mandatory folic acid fortification prevent or promote cancer?” American Journal of Clinical Nutrition, 2004; 80(5): 1123-1128. A meta-analysis of randomized controlled trials of B vitamin supplementation (which included folic acid) in cardiovascular disease patients found no significant increase in cancer incidence or mortality, but the follow-up periods were relatively short for cancer outcomes.
The unmetabolized folic acid (UMFA) question remains open. UMFA appears in the bloodstream at folic acid intakes above approximately 200-300 μg and is detectable in a significant portion of the U.S. population since fortification began. Whether circulating UMFA has biological effects (such as competing with 5-MTHF for folate receptors or transporters) or is simply an inert byproduct of excess intake is not settled. The absence of demonstrated harm does not constitute evidence of safety at population scale, and this uncertainty is one of the reasons many European countries have resisted mandatory fortification.
Dietary sources and requirements
Good dietary sources of natural folate include dark leafy greens (spinach, kale, romaine lettuce), legumes (lentils, chickpeas, black beans), asparagus, broccoli, Brussels sprouts, avocado, citrus fruits, and liver. Natural food folates are heat-sensitive and water-soluble, so cooking losses can be substantial.
The RDA for adults is 400 μg DFE/day. For women capable of becoming pregnant, the additional recommendation is 400 μg of folic acid specifically (not food folate alone) from supplements or fortified foods, on top of dietary folate, to achieve NTD-protective levels. Pregnancy increases the RDA to 600 μg DFE/day, and lactation to 500 μg DFE/day.14Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: National Academies Press, 1998.
The tolerable upper intake level of 1,000 μg/day applies to folic acid from supplements and fortified foods only, not to food folate, because the masking of B12 deficiency has only been demonstrated with synthetic folic acid. There is no established UL for food folate or for 5-MTHF supplementation, though the same caution about B12 masking logically applies to any folate form that corrects megaloblastic anemia.
Who needs to supplement
Women of childbearing age have the strongest and most time-sensitive case for supplementation. Because the neural tube closes before most women know they are pregnant, and because half of pregnancies are unplanned, supplementation should be ongoing in any woman who might become pregnant. The recommended dose is 400-800 μg of folic acid (or equivalent 5-MTHF) daily. Women with a prior NTD-affected pregnancy should take 4 mg/day under medical supervision.
People with malabsorption conditions, including celiac disease, inflammatory bowel disease, and short bowel syndrome, may not absorb dietary folate efficiently and may benefit from supplementation.
People taking medications that interfere with folate metabolism have a clear supplementation rationale. Methotrexate inhibits DHFR directly (this is its mechanism of action as a chemotherapy and immunosuppressive agent). Certain anticonvulsants (phenytoin, carbamazepine, valproic acid) increase folate catabolism. Sulfasalazine inhibits folate absorption. In each case, folate status should be monitored and supplemented as needed.
Heavy alcohol users are at risk for folate depletion through multiple mechanisms: reduced dietary intake, impaired intestinal absorption, increased urinary excretion, and direct inhibition of folate-dependent enzymes by acetaldehyde.
People with elevated homocysteine may benefit from folate supplementation (often combined with B12 and B6) to lower levels, though as discussed in the B vitamins overview, the evidence that this lowers cardiovascular event rates is weak.
For the general population eating a varied diet in a country with mandatory folic acid fortification, folate deficiency is uncommon. The groups most likely to have inadequate status are those with restricted diets, limited access to fortified foods, heavy alcohol use, or medication-related folate depletion.
Conclusion
Folate sits at the center of one-carbon metabolism, supplying the carbon units for DNA synthesis and the methyl groups for the entire SAM-dependent methylation system. Its role in neural tube defect prevention is the strongest and most consequential supplement recommendation in nutrition. The folate cycle’s connection to thymidylate synthesis, purine synthesis, and homocysteine remethylation means that inadequate folate status affects DNA integrity, cell division, and methylation simultaneously.
The choice between folic acid and 5-MTHF as a supplement is a practical question with a nuanced answer. Folic acid has the clinical evidence base for NTD prevention, the longest track record, the greatest stability, and the lowest cost. 5-MTHF bypasses both DHFR and MTHFR, avoids the generation of unmetabolized folic acid, and is a reasonable choice for people concerned about MTHFR polymorphisms or high-dose folic acid exposure. For most people at recommended doses, both forms effectively raise folate status. The most important variable is not the form but whether supplementation is happening at all, particularly in women before and during early pregnancy.
Generate Your Stack. Avoid Conflicts. Optimize Absorption.
Most supplement advice ignores timing, interactions, and dose-response curves. Smart Stack Builder doesn't. Get a personalized daily schedule with interaction flags and dosage guidance, built on the same evidence base as this article.
References
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
This article is part of our Supplements hub: A science-backed collection of ingredient research, clinical dosage reviews, and optimized stack insights.