Vitamin B12 (cobalamin)
Vitamin B12
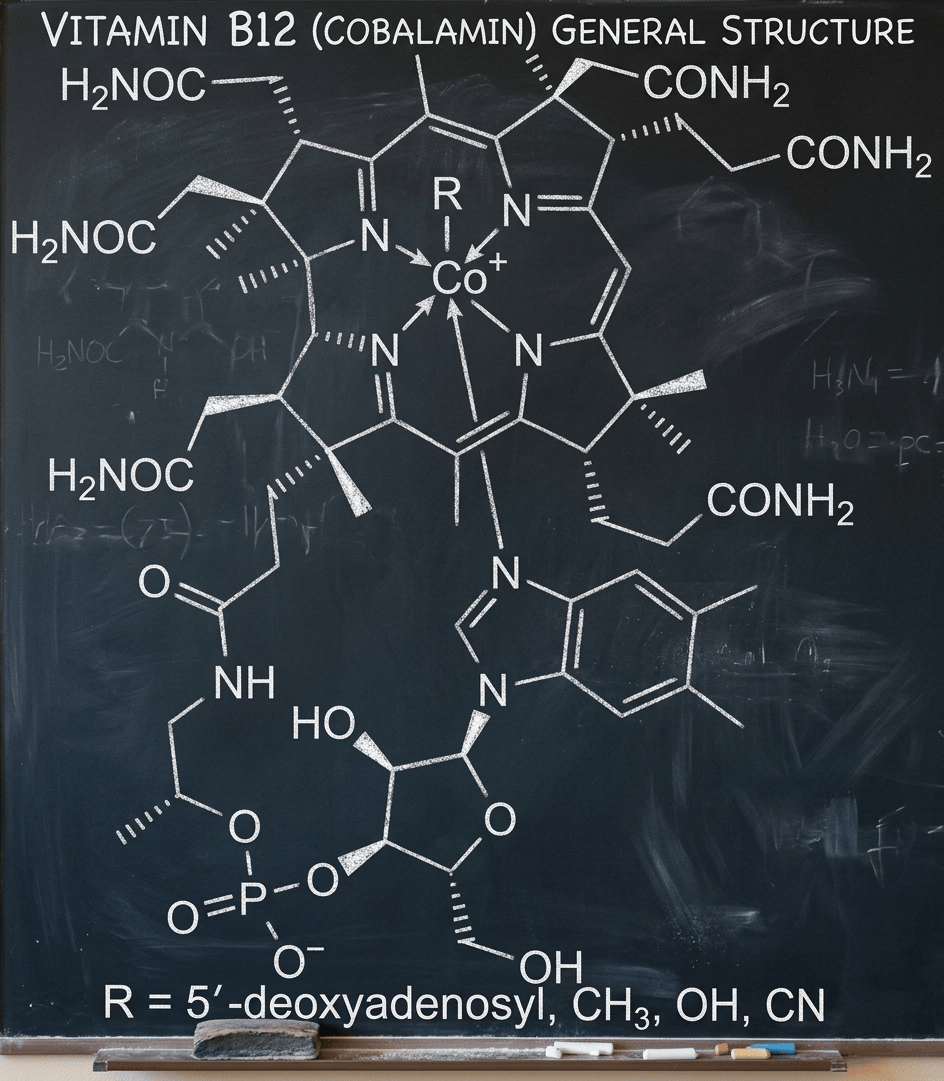
Vitamin B12 is the largest and most structurally complex of all the vitamins. Its core is a corrin ring, a structure similar to the porphyrin ring in hemoglobin, with a cobalt atom at the center. That cobalt atom is where the chemistry happens: it can form bonds with different upper ligands, and which ligand is attached determines which form of B12 you are dealing with. Cyanocobalamin has a cyanide group. Hydroxocobalamin has a hydroxyl group. Methylcobalamin has a methyl group. Adenosylcobalamin (also called 5′-deoxyadenosylcobalamin) has an adenosyl group. The first two are the forms found in supplements and pharmaceuticals. The last two are the forms that actually function as coenzymes in human metabolism.
The body uses B12 for exactly two enzymatic reactions. That sounds underwhelming compared to the 140+ reactions that depend on vitamin B6, but these two reactions sit at metabolic chokepoints that affect DNA synthesis, red blood cell production, myelin maintenance, and the entire one-carbon metabolism system. When B12 is insufficient, the damage is slow, systemic, and in some cases irreversible.
The B vitamins overview on this site covers B12’s role in one-carbon metabolism, the methyl trap, and the homocysteine connection. This article goes deeper into how B12 gets absorbed (a process more complicated than any other vitamin), the clinical consequences of deficiency, the differences between supplemental forms, and who genuinely needs to supplement.
A necessary disclaimer: If you have been diagnosed with pernicious anemia, have had gastric or ileal surgery, or are experiencing neurological symptoms such as numbness, tingling, or balance problems, work with a qualified medical professional. B12 deficiency can cause permanent neurological damage if left untreated. This article is educational. It is not a treatment plan, a diagnostic tool, or a substitute for clinical care.
The two reactions
Methionine synthase
Methylcobalamin is the cofactor for methionine synthase, the enzyme that converts homocysteine to methionine. In this reaction, the methyl group from 5-methyltetrahydrofolate (5-mTHF) is transferred to cobalamin, which then passes it to homocysteine, regenerating methionine and simultaneously converting 5-mTHF back to tetrahydrofolate (THF).1Banerjee, R. and Ragsdale, S.W. “The Many Faces of Vitamin B12: Catalysis by Cobalamin-Dependent Enzymes.” Annual Review of Biochemistry, 2003; 72: 209-247.
This single reaction connects three metabolic systems. It regenerates methionine, which is required for the production of S-adenosylmethionine (SAM), the body’s universal methyl donor for DNA methylation, neurotransmitter synthesis, and myelin maintenance. It recycles THF, which is necessary for de novo nucleotide synthesis and therefore DNA replication. And it clears homocysteine, preventing its accumulation. When B12 is deficient, all three systems are impaired simultaneously: SAM production drops, THF becomes trapped as 5-mTHF (the “methyl trap” described in the B vitamins overview), and homocysteine accumulates.
The methyl trap is the reason B12 deficiency and folate deficiency produce identical hematological symptoms. Both cause megaloblastic anemia because both result in insufficient THF for DNA synthesis. But only B12 deficiency causes neurological damage, because the neurological consequences stem from SAM depletion and its effects on myelin, not from the folate-dependent nucleotide pathway. This distinction has a dangerous clinical implication: high-dose folate supplementation can correct the anemia of B12 deficiency by partially bypassing the trapped folate, making blood counts look normal while neurological damage progresses undetected.2Stabler, S.P. “Vitamin B12 Deficiency.” New England Journal of Medicine, 2013; 368(2): 149-160.
Methylmalonyl-CoA mutase
Adenosylcobalamin is the cofactor for methylmalonyl-CoA mutase, a mitochondrial enzyme that converts methylmalonyl-CoA to succinyl-CoA. This reaction is part of the catabolic pathway for odd-chain fatty acids, branched-chain amino acids (valine, isoleucine), and the side chain of cholesterol. When this reaction fails due to B12 deficiency, methylmalonic acid (MMA) accumulates in blood and urine.3Banerjee, R. and Ragsdale, S.W. “The Many Faces of Vitamin B12: Catalysis by Cobalamin-Dependent Enzymes.” Annual Review of Biochemistry, 2003; 72: 209-247.
Elevated MMA is the most specific biomarker for B12 deficiency. Homocysteine can be elevated by deficiencies in B6, folate, or B12, but MMA rises only in B12 deficiency (or in rare genetic conditions). This makes the combination of elevated MMA and elevated homocysteine a strong diagnostic indicator for B12 insufficiency, even when serum B12 levels fall within the conventional normal range.
The methylmalonyl-CoA mutase pathway also has a less commonly discussed connection to neurological function. Accumulation of methylmalonic acid and its precursor, methylmalonyl-CoA, can disrupt normal fatty acid synthesis. Methylmalonyl-CoA competes with malonyl-CoA as a substrate for fatty acid synthase, potentially leading to the incorporation of abnormal branched-chain fatty acids into myelin lipids. This is one proposed mechanism for the demyelination seen in B12 deficiency, distinct from and additive to the SAM depletion pathway.4Stabler, S.P. “Vitamin B12 Deficiency.” New England Journal of Medicine, 2013; 368(2): 149-160.
How B12 gets absorbed
B12 absorption is a multi-organ relay unlike anything else in vitamin metabolism. It requires the stomach, the pancreas, and the terminal ileum, each contributing a different step. Failure at any point in the chain can cause deficiency regardless of dietary intake.
In the stomach, hydrochloric acid and pepsin separate B12 from the food proteins it is bound to. The freed B12 immediately binds to haptocorrin (also called R-protein or transcobalamin I), a glycoprotein secreted by the salivary glands and gastric mucosa. Haptocorrin protects B12 from the acidic stomach environment but is not involved in absorption itself.
In the duodenum, pancreatic proteases degrade haptocorrin, releasing B12 again. The freed B12 then binds to intrinsic factor, a glycoprotein produced by the stomach’s parietal cells. This binding occurs at a higher pH and is specific: intrinsic factor binds cobalamin with high affinity but not other corrinoids.5Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: National Academies Press, 1998.
The intrinsic factor-B12 complex travels to the terminal ileum, where it binds to a receptor called cubam (a complex of cubilin and amnionless) on the brush border of ileal enterocytes. The complex is internalized by receptor-mediated endocytosis. Inside the enterocyte, intrinsic factor is degraded in lysosomes and B12 is released, then bound to transcobalamin II for transport into the circulation. From there it is delivered to cells throughout the body.
This intrinsic factor-dependent pathway has a limited capacity: it can absorb roughly 1.5-2 μg of B12 per meal.6Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: National Academies Press, 1998. A second absorption pathway exists: passive diffusion across the intestinal mucosa, which is independent of intrinsic factor but accounts for only about 1-2% of the dose. At physiological doses, passive diffusion is negligible. At pharmacological doses (1,000 μg or higher), it becomes clinically relevant, which is why high-dose oral B12 can treat deficiency even in people who lack intrinsic factor.
The liver stores 2-5 mg of B12, and the vitamin is efficiently conserved through enterohepatic circulation: B12 secreted in bile is reabsorbed in the ileum. These reserves mean that deficiency from dietary insufficiency alone takes years to develop, typically 3-5 years for strict vegans, sometimes longer. Deficiency from malabsorption (loss of intrinsic factor, ileal disease, or gastric surgery) can develop faster because both dietary B12 and the enterohepatic pool are affected.
What deficiency looks like
B12 deficiency manifests in two systems that can be affected independently: the hematological system and the nervous system.
Hematological effects
The hallmark is megaloblastic anemia, caused by impaired DNA synthesis in rapidly dividing cells. Red blood cell precursors in the bone marrow grow larger than normal because they cannot replicate their DNA efficiently, producing the characteristic large, oval-shaped red cells (macro-ovalocytes) and hypersegmented neutrophils visible on a peripheral blood smear. Mean corpuscular volume (MCV) rises above 100 fL, and values above 115 fL are particularly suggestive of B12 or folate deficiency. The anemia can be severe, and pancytopenia (depression of all blood cell lines) can occur in advanced cases.
Neurological effects
The neurological damage from B12 deficiency can occur without anemia, which is part of what makes it dangerous. The classic presentation is subacute combined degeneration of the spinal cord: demyelination of the dorsal columns (causing loss of vibration sense and proprioception) and the lateral corticospinal tracts (causing weakness and spasticity). Peripheral neuropathy with paresthesias in the hands and feet is common. Cognitive symptoms, including memory loss, confusion, irritability, and depression, can appear early and may be the only presenting signs.7Stabler, S.P. “Vitamin B12 Deficiency.” New England Journal of Medicine, 2013; 368(2): 149-160.
The neurological damage progresses independently of the hematological effects because the mechanisms are different. The anemia results from failed DNA synthesis (the folate-dependent pathway). The neurological damage results from failed methylation (the SAM-dependent pathway) and possibly from abnormal fatty acid incorporation into myelin (the methylmalonic acid pathway). Treatment within weeks of symptom onset typically produces full recovery. Delayed treatment allows damage to become permanent. In one review, 86% of patients with subacute combined degeneration showed clinical improvement after B12 replacement, but only 14% achieved complete resolution.8Kumar, N. “Neurologic Aspects of Cobalamin (B12) Deficiency.” Handbook of Clinical Neurology, 2014; 120: 915-926.
Diagnostic pitfalls
Serum B12 levels are an imperfect screening tool. The conventional reference range (200-900 pg/mL) includes a substantial gray zone: functional deficiency can exist at levels that appear normal, particularly in the 200-400 pg/mL range. Elevated MMA and homocysteine are more sensitive indicators. Anti-intrinsic factor antibodies can cause falsely normal or even falsely elevated serum B12 readings in some assays, meaning that a normal B12 level does not exclude pernicious anemia in patients with compatible symptoms. When clinical suspicion is high, MMA and homocysteine should be measured regardless of the serum B12 result.
Supplemental forms
Four forms of B12 are commercially available: cyanocobalamin, hydroxocobalamin, methylcobalamin, and adenosylcobalamin. Understanding what each one is (and is not) helps cut through the marketing noise.
Cyanocobalamin is synthetic. It does not occur in meaningful quantities in food or in the body. It is the most stable form, the cheapest to produce, and the most extensively studied in clinical trials. Once absorbed, the body removes the cyanide group (the amount is toxicologically trivial) and converts cyanocobalamin to methylcobalamin and adenosylcobalamin for use. It is the form used in most fortified foods, most multivitamins, and most clinical research on B12 supplementation.9Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: National Academies Press, 1998.
Hydroxocobalamin is the form predominantly found in food. It is also the form used for B12 injections in many countries outside the United States. After injection, hydroxocobalamin is retained in the body longer than cyanocobalamin, requiring less frequent dosing. It is also used as an antidote for cyanide poisoning at very high intravenous doses.
Methylcobalamin is one of the two active coenzyme forms (the cofactor for methionine synthase). It is widely marketed as the “natural” or “active” form and is popular in supplements. The premise is that it bypasses a conversion step and is therefore more bioavailable or more effective.
Adenosylcobalamin is the other active coenzyme form (the cofactor for methylmalonyl-CoA mutase). It is less commonly sold as a standalone supplement but appears in some combination products.
The evidence on whether the form matters for most people is less definitive than supplement marketing implies. A 1971 study found that cyanocobalamin, hydroxocobalamin, methylcobalamin, and adenosylcobalamin were all absorbed at similar rates at physiological doses.10Adams, J.F., Ross, S.K., Mervyn, L., Boddy, K., and King, P. “Absorption of cyanocobalamin, coenzyme B12, methylcobalamin, and hydroxocobalamin at different dose levels.” Scandinavian Journal of Gastroenterology, 1971; 6(3): 249-252. Cyanocobalamin may be absorbed slightly better but is excreted more rapidly in urine; methylcobalamin appears to be retained somewhat longer in tissues.11Okuda, K., Yashima, K., Kitazaki, T., and Takara, I. “Intestinal absorption and concurrent chemical changes of methylcobalamin.” Journal of Laboratory and Clinical Medicine, 1973; 81(4): 557-567. A study in vegans found that cyanocobalamin produced higher holotranscobalamin levels (the bioactive fraction of circulating B12) than methylcobalamin at comparable doses.12Zugravu, C.A., Macri, A., Belc, N., and Bohiltea, R. “Efficacy of supplementation with methylcobalamin and cyanocobalamin in maintaining the level of serum holotranscobalamin in a group of plant-based diet (vegan) adults.” Experimental and Therapeutic Medicine, 2021; 22(3): 993. The NIH Office of Dietary Supplements states that there is no evidence that absorption rates of supplemental B12 vary by form.
Two important caveats. First, the body needs both methylcobalamin and adenosylcobalamin to function. Taking only methylcobalamin provides only one of the two coenzyme forms; the body must still convert some of it to adenosylcobalamin, which requires the same enzymatic machinery that processes cyanocobalamin. The “no conversion needed” claim for methylcobalamin is therefore only half true. Second, methylcobalamin is less stable than cyanocobalamin and more sensitive to light degradation, which can affect product quality depending on storage conditions.
For most people, the form of B12 matters less than whether they are getting enough. Cyanocobalamin has the longest track record, the most clinical evidence, and the lowest cost. Methylcobalamin and adenosylcobalamin are reasonable alternatives. Hydroxocobalamin is the preferred form for injections due to its longer retention time. Anyone with a specific concern about B12 metabolism (such as known MTHFR polymorphisms, renal insufficiency, or cyanide exposure from smoking) may have reason to prefer one form over another, but for the general population, the differences are not clinically meaningful at adequate doses.
Oral vs. intramuscular supplementation
The traditional teaching was that B12 deficiency, particularly from pernicious anemia, requires lifelong intramuscular injections because oral absorption depends on intrinsic factor. This is outdated.
The passive diffusion pathway absorbs approximately 1-2% of an oral dose regardless of intrinsic factor status. At a dose of 1,000 μg, that means roughly 10-20 μg reaches the bloodstream through passive absorption alone, far exceeding the 2.4 μg RDA. A Cochrane review of randomized controlled trials found that 1,000 μg/day of oral B12 was equivalent to intramuscular B12 for normalizing serum levels and achieving hematological responses, and that 2,000 μg/day oral B12 may be superior.13Wang, H., Li, L., Qin, L.L., Song, Y., Vidal-Alaball, J., and Liu, T.H. “Oral vitamin B12 versus intramuscular vitamin B12 for vitamin B12 deficiency.” Cochrane Database of Systematic Reviews, 2018; 3: CD004655.
A 2020 noninferiority trial in adults over 65 found no significant difference between oral and intramuscular cyanocobalamin in normalizing B12 levels at 8, 26, or 52 weeks. A 2023 network meta-analysis comparing oral, sublingual, and intramuscular routes concluded that all three effectively increased serum B12 with no significant differences between them.
Injections retain a role for severe neurological presentations (where clinicians may prefer the certainty of bypassing the gut entirely), for patients who cannot reliably take oral medications, and in clinical settings where adherence is a concern. But for most cases of B12 deficiency, high-dose oral supplementation (1,000-2,000 μg/day) is effective, less expensive, and better tolerated than injections.
Who needs to supplement
The RDA for B12 is 2.4 μg/day for adults. Because B12 is found almost exclusively in animal-derived foods, the populations at genuine risk for deficiency are predictable:
Vegans and strict vegetarians have the most straightforward case for supplementation. B12 is not produced by plants. Fermented foods, algae, and nutritional yeast are sometimes cited as plant sources, but the B12 content is unreliable, and some contains B12 analogues that may actually interfere with true B12 metabolism. Supplementation or regular consumption of B12-fortified foods is considered essential for anyone following a plant-based diet long-term.14Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: National Academies Press, 1998.
Adults over 50 are at increased risk because of age-related declines in gastric acid production, which impairs the release of food-bound B12 in the stomach. The Institute of Medicine specifically recommends that adults over 50 obtain their B12 from fortified foods or supplements, because crystalline B12 (the form in supplements and fortified foods) does not require gastric acid for absorption. Atrophic gastritis, which affects an estimated 10-30% of older adults, compounds this problem.
People with pernicious anemia produce autoantibodies against intrinsic factor or parietal cells, eventually losing the ability to absorb B12 through the intrinsic factor pathway. This is typically a lifelong condition requiring ongoing supplementation, either by injection or by high-dose oral B12.
People who have had gastric or ileal surgery, including gastric bypass, sleeve gastrectomy, or ileal resection for Crohn’s disease, may have permanently impaired B12 absorption depending on the extent of the surgery.
People taking certain medications are at risk. Metformin reduces B12 absorption through a mechanism that is not fully understood but may involve altered intestinal motility or changes in gut bacteria. Proton pump inhibitors and H2 receptor antagonists reduce gastric acid secretion, impairing the release of food-bound B12. Long-term use of either drug class is associated with lower B12 status.
Chronic nitrous oxide exposure deserves special mention. Nitrous oxide irreversibly oxidizes the cobalt atom in B12, inactivating the vitamin. A single prolonged exposure (such as recreational use or extended surgical anesthesia) can precipitate acute B12-deficient neurological symptoms in people with marginal B12 stores.
Safety
B12 has no established tolerable upper intake level because oral toxicity has not been demonstrated at any dose studied. The IOM concluded that there was insufficient evidence of harm to set a UL.15Institute of Medicine. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline. Washington, DC: National Academies Press, 1998. Excess B12 is excreted in urine. Allergic reactions to injectable B12 have been reported rarely.
Some epidemiological studies have found associations between high-dose B12 supplementation and increased lung cancer risk, particularly in male smokers, but the evidence is observational, the mechanism is unclear, and no causal relationship has been established. This is worth monitoring but is not a basis for avoiding supplementation in people who need it.
Conclusion
B12 deficiency is common and underdiagnosed, and when it affects the nervous system, potentially irreversible. The absorption pathway is uniquely vulnerable to disruption at multiple points, which is why deficiency occurs not only in people with inadequate intake but also in people with normal diets who have impaired gastric acid production, autoimmune destruction of parietal cells, or ileal disease. Serum B12 levels can be misleadingly normal in the presence of functional deficiency, making MMA and homocysteine important confirmatory tests when clinical suspicion is high.
For supplementation, the form of B12 matters less than the dose and consistency. Cyanocobalamin has the most evidence, the best stability, and the lowest cost. Methylcobalamin and adenosylcobalamin are the active coenzyme forms but require adequate doses and proper storage. High-dose oral supplementation (1,000-2,000 μg/day) is effective even in pernicious anemia, thanks to the passive diffusion pathway that bypasses intrinsic factor. Injections remain appropriate for acute neurological presentations and for patients who cannot reliably take oral supplements.
The populations with clear supplementation needs are vegans (essential), adults over 50 (recommended by the IOM), people with pernicious anemia or post-surgical malabsorption (medically necessary), and people on long-term metformin or acid-suppressing medications (worth monitoring and supplementing if status declines). For everyone else, a varied diet that includes animal products typically provides adequate B12 without supplementation.
Generate Your Stack. Avoid Conflicts. Optimize Absorption.
Most supplement advice ignores timing, interactions, and dose-response curves. Smart Stack Builder doesn't. Get a personalized daily schedule with interaction flags and dosage guidance, built on the same evidence base as this article.
References
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
- 15
This article is part of our Supplements hub: A science-backed collection of ingredient research, clinical dosage reviews, and optimized stack insights.